-
挥发性有机物(volatile organic compounds,VOCs)大多数具有剧毒、致癌和危险性[1],同时也是区域臭氧和二次有机气溶胶形成的主要原因[2],严重影响人类健康和大气环境。我国工业源排放的VOCs占人为排放总量的60%左右[3],其中化工、工业涂装、印刷和家具制造占中国总排放量的50%,十四五规划纲要建议强化大气污染综合治理和联防联控,对环境空气质量提出更高要求,因此,应当对工业源VOCs排放加强综合控制。
印刷业VOCs排放具有风量大、浓度低等特点,主要通过吸附法进行末端处理。工业应用中常见的吸附剂主要有活性炭、分子筛、树脂等[4–6],活性炭因具有比表面积大、孔隙率高以及稳定性好等特点被广泛应用[7]。在吸附法治理VOCs废气中,废气由多组分VOCs组成,吸附净化过程中会产生竞争吸附现象。HUANG等[8]发现饱和蒸气压较低的VOCs会取代饱和蒸气压较高的VOCs。YAO等[9]发现CAR-AC吸附二元VOCs时,乙酸乙酯对丙酮和乙醛产生竞争现象,导致丙酮和乙醛产生“驼峰”,使其在活性炭上的吸附容量降低。有研究表明,VOCs吸附容量与活性炭的活性位点有关[10],目前增加活性位点的方法分为物理改性和化学改性,物理改性主要是在高温下活化条件下增加比表面积和孔体积[11]。化学改性有酸改性、碱改性、盐改性、杂原子改性和掺杂金属氧化物等[12-14]。相较于其他方法,掺杂金属氧化物方法具有环境污染小、对特定的VOCs亲和力高等特点。ZHOU等[15]通过浸渍法将Mg、Zn、Cu和Zr掺杂进活性炭中,发现AC/ZnO对丙酮和甲醇的吸附性能最好;金春江等[16]通过对山桃核基活性炭掺杂Fe(NO3)3制备的AC-3对乙酸乙酯的吸附容量从498.07 mg·g−1提升至973.04 mg·g−1,YANG[17]在生物质炭中掺杂氧化镁以增强对极性VOCs的吸附性能。BAUR等[18]通过向活性炭纤维中掺杂碱性金属氧化物(La2O3、CaO、MgO、ZnO、Al2O3)为乙醛吸附提供了更多的活性位点。目前改性活性炭提高对极性VOCs的吸附研究多集中在单一组分,二元吸附研究较少。
本研究根据彩板印刷排放特征,选取甲苯、乙酸乙酯和异丙醇为特征污染物,采用等体积浸渍法制备出针对极性VOCs吸附容量强的载镁活性炭,并通过实验进行二元吸附性能测试,结合表征和密度泛函理论从微观层面分析竞争吸附机制,为活性炭吸附VOCs的工业应用提供参考。
-
活性炭购置于江苏省常州碳家乐煤基柱状活性炭,粒径4 mm,碘值为800 mg·g−1;甲苯、乙酸乙酯、异丙醇购置于阿拉丁生化科技有限公司,均为分析纯,其分子量、密度等相关物性参数见表1。氯化镁购置于天力化学试剂有限公司。
-
将煤基活性炭在105 ℃恒温条件下干燥12 h(原始活性炭命名为AC)。以MgCl2溶液为金属前驱体,用去离子水配出不同质量分数的MgCl2溶液,采用等体积浸渍法取5 g经预处理后的煤基活性炭于MgCl2溶液中浸渍12 h,取出在105 ℃恒温干燥12 h,将干燥后的活性炭置于马弗炉中,在空气条件下以5 ℃·min−1的速率将温度升至450 ℃并保温2.5 h,自然冷却至室温即制得载镁活性炭,将负载氧化镁质量分数分别为0.5%、1%、1.5%、2%的样品命名为MgO/AC-0.5%、MgO/AC-1%、MgO/AC-1.5%及MgO/AC-2%。未掺杂氧化镁同等条件下处理的活性炭命名为MgO/AC-0%。焙烧改性实验:氧化镁掺杂质量分数为1%,在不同焙烧温度350、400、450、500 ℃下进行样品制备。
-
吸附实验在常温常压下进行,实验系统如图1所示。由注射泵(LSP02-D)将VOCs液体注入三颈烧瓶内并置于恒温水浴锅(50 ℃)中,经硅胶除湿后的空气由质量流量计(北京七星CS-200D,10 L)控制流量为3 L·min−1进入三颈烧瓶中,经缓冲瓶稳定VOCs废气质量浓度后,进入填装活性炭的吸附器(内径为10 mm、装填高度为120 mm,装填质量3.20 g)吸附后排出。吸附器进、出口VOCs质量浓度使用气相色谱仪(福立GC9700Ⅱ)测定,当吸附器VOCs出口质量浓度为进口质量浓度的95%时认为吸附饱和,结束实验,尾气经净化后排出。
以吸附时间t为横坐标,以t min时出口和入口VOCs质量浓度之比(Ct/C0)为纵坐标,得到VOCs的吸附穿透曲线。通过式(1)对吸附穿透曲线进行积分计算,得到VOCs的平衡吸附容量。
式中:Q为平衡吸附容量,mg·g−1;F为气流速度,L·min−1;C0为进口VOCs质量浓度,mg·m−3;Ct为t 时刻吸附器出口VOCs质量浓度,mg·m−3;W为吸附剂装填量,g;ts为吸附平衡时间,min。
-
采用扫描电子显微镜(Gemini500)对活性炭结构进行表征;采用能量色散光谱(JEOL Corp)对活性炭中的所有元素种类和含量进行测定;采用比表面积测试仪(V-Sorb 2800P)对活性炭比表面积孔径分布进行表征;采用傅里叶红外光谱仪(Nicolet Antaris IGS)对活性炭表面官能团进行表征;采用X射线衍射仪(UItima Ⅳ)对活性炭结构进行表征。
-
吸附力强弱通过吸附能判断,采用VASP软件计算,其中VASP计算方法基于赝势平面波基组,电子交换关联势采用广义梯度近似法(generalized gradient approximation,GGA)中的Perdew-Burke-Ernzerhof(PBE)泛函。对模型k点的网格大小为1×1×1,平面波展开的截断能为400 eV。强制k网格以gamma点为中心。使用宽度为0.1 eV的高斯展宽。电子迭代收敛性设置为1×10−5 eV以及倒数空间投影算子,原子力收敛判据设置为0.02 eV·Å−1。吸附能根据式(2)进行计算。
式中:E为吸附能,kJ·mol−1;
$ {{E}}_{\text{AC+gas}} $ 为吸附质在吸附剂吸附时体系的总能量,kJ·mol−1;$ {{E}}_{\text{AC}} $ 为吸附剂的能量,kJ·mol−1;$ {{E}}_{\text{gas}} $ 为吸附质的能量,kJ·mol−1。吸附能表示吸附后体系的稳定性,吸附能越低,表明体系越稳定,越不容易被强吸附质取代。 -
1)氧化镁掺杂量对活性炭吸附性能的影响。对AC、AC/MgO-0%、AC/MgO-0.5%、AC/MgO-1%、AC/MgO-1.5%和AC/MgO-2%分别进行甲苯、乙酸乙酯、异丙醇的吸附性能实验,结果如图2所示。图2可以看出,甲苯、乙酸乙酯、异丙醇吸附穿透曲线随着氧化镁掺杂量提高先向右移再向左移,其中AC/MgO-1%对甲苯和乙酸乙酯的穿透曲线位于最右侧,AC/MgO-0%对异丙醇的穿透曲线位于最右侧,但上升较快,考虑可能是未掺杂氧化镁导致其介孔提高,使得对异丙醇的传质速率加快,吸附初期穿透曲线上升缓慢,在吸附后期其微孔体积减少导致VOCs的容量减少。AC/MgO-1%对3种VOCs的穿透时间分别延长了40、30、30 min。随着掺杂量的升高,AC/MgO对甲苯、乙酸乙酯、异丙醇的吸附容量出现先增加后减少的趋势,AC/MgO-1%对甲苯、乙酸乙酯、异丙醇的吸附容量最大,分别由229、148、97 mg·g−1提高至326 (提高了42%)、175 (提高了18%)、124 mg·g−1(提高了28%)。这可能由于掺杂MgO后的AC中增加了对乙酸乙酯和异丙醇的极性吸附位点,从而使得其吸附容量提高;另外,AC/MgO-0%相对于原始AC,吸附容量分别提高了28%、16%、24%,高温焙烧使得活性炭的石墨化程度提高[19],因此,氧化镁最佳掺杂量为1%。
2)焙烧温度对改性活性炭吸附性能影响。不同焙烧温度下制备的AC/MgO-1%改性活性炭对VOCs吸附性能的影响如图3所示。由图3可以看出,随着焙烧温度的增加,甲苯吸附穿透曲线均向右偏移,吸附容量逐渐增大,而乙酸乙酯和异丙醇的吸附穿透曲线先向右偏移后再向左偏移,吸附容量则是先增大后减小。500 ℃时焙烧制备的掺杂改性活性炭对甲苯的吸附容量最大,而450 ℃焙烧制备的掺杂改性活性炭对乙酸乙酯和异丙醇吸附容量最大。原因可能是随着焙烧温度升高,活性炭的石墨化程度随之提高,对甲苯的吸附容量增大;而负载氧化镁会增加对乙酸乙酯和异丙醇的吸附位点,从而使其对乙酸乙酯和异丙醇的吸附容量增加。综合考虑甲苯、乙酸乙酯和异丙醇的吸附容量,选择最佳焙烧温度为450 ℃。
-
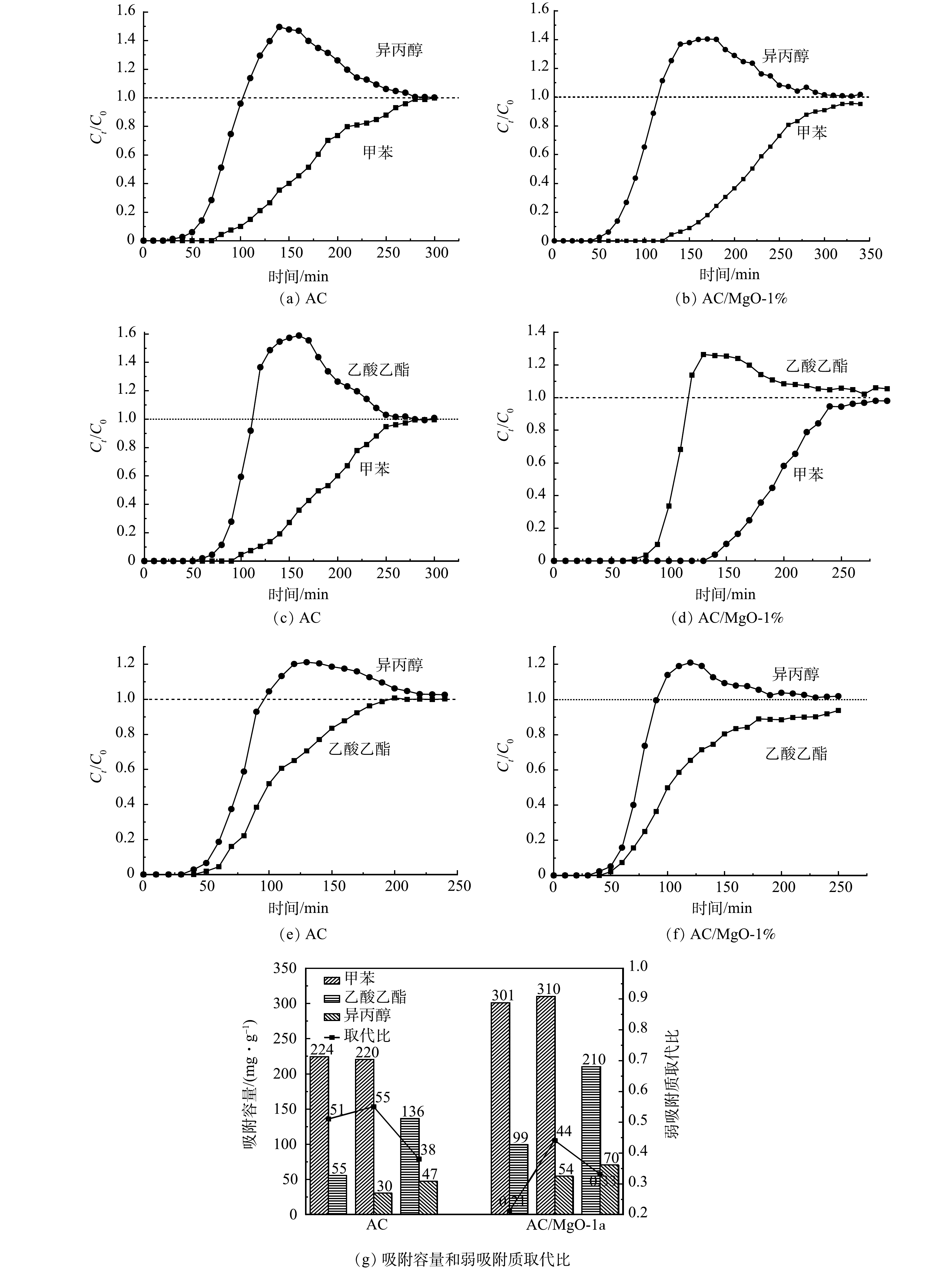
双组分吸附性能可通过穿透时间、吸附容量及“驼峰”面积评价,分别对AC和AC/MgO-1%进行甲苯、乙酸乙酯及异丙醇的二元吸附实验,结果如图4所示。由图4(a)~(f)可以看出,双组分VOCs在吸附过程依次经历3个阶段:共吸附阶段、竞争吸附阶段和平衡吸附阶段。在共吸附阶段,由于活性炭上的吸附位点未被占据,强吸附质和弱吸附质均等地被吸附到吸附位点上。在竞争吸附阶段,活性炭的吸附位点被大量占据,弱吸附质由于和活性炭的结合较弱,被强吸附质取代,导致弱吸附质从活性炭中脱附出来,其穿透曲线出现“驼峰”。由于AC属于非极性吸附剂,其对弱极性甲苯的吸附作用比极性吸附质乙酸乙酯和异丙醇强,且甲苯的沸点高于乙酸乙酯和异丙醇,甲苯相比于乙酸乙酯和异丙醇均为强吸附质。因此,图4(a)、图4(b)中对于甲苯-异丙醇二元吸附,吸附穿透曲线均出现异丙醇的“驼峰”;而图4(c)、图4(d)中对于甲苯-乙酸乙酯二元吸附,吸附穿透曲线均出现乙酸乙酯的“驼峰”。而对于极性指数相同的吸附质乙酸乙酯和异丙醇二元体系,虽然异丙醇沸点高于乙酸乙酯,但竞争吸附并未遵循一般的高沸点置换低沸点规律,考虑孔堵塞效应所致[20],如乙酸乙酯动力学直径大于异丙醇,因此,图4(e)、图4(f)中对于乙酸乙酯-异丙醇二元吸附,吸附穿透曲线均出现异丙醇的“驼峰”。在平衡吸附阶段,弱吸附质和强吸附质出口质量浓度均接近初始质量浓度C0(Ci/C0=1),吸附达到平衡。
同时可以发现,相比AC,掺杂氧化镁改性的AC/MgO-1%吸附甲苯-乙酸乙酯和甲苯-异丙醇时,穿透时间均延长、“驼峰”降低;而对于乙酸乙酯-异丙醇,“驼峰”下降不明显,考虑由乙酸乙酯和异丙醇的理化性质相似导致。将二元吸附过程中弱吸附质竞争吸附阶段的取代量与总吸附容量的比值定义为弱吸附质被取代比,其中,弱吸附质的总吸附量和竞争吸附阶段被取代量(驼峰部分)通过式(1)计算,结果如图4(g)所示。由图4(g)可以看出,掺杂氧化镁改性后的活性炭吸附容量较AC均有提高(34%~80%),而二元吸附时弱吸附质的被取代占比均有下降(21%~33%),说明竞争作用减弱。
-
以甲苯、乙酸乙酯、异丙醇为吸附质,对性能最佳的AC/MgO-1%样品进行了多次吸脱附实验,吸附实验同前,脱附实验在空气气氛下温度为200 ℃脱附2 h,后对样品进行动态吸附脱附循环实验,以吸附量和循环使用率(脱附后的VOCs吸附量与首次使用的吸附量的比值)表征吸附剂的稳定性,结果如图5所示。如图5所示,AC/MgO-1%经5次吸附脱附循环实验后,吸附容量稍有下降。其中,甲苯由326 mg·g−1降至302 mg·g−1,降低了7.36%;乙酸乙酯由175 mg·g−1降至168 mg·g−1,降低了4.00%;异丙醇由124 mg·g−1降至118 mg·g−1,降低了4.84%。这表明AC/MgO-1%的吸附性能较为稳定。同时,经5次吸附脱附循环实验后,使用率仍保持在92.68%以上,这表明经过改性的AC/MgO-1%再生性能良好。
-
1) XRD谱图分析。AC、AC/MgO-0%和AC/MgO-1%的XRD谱图结果如图6所示。由图6可以看出,AC在25°和43°处有2个衍射峰分别对应于六方石墨的(002)和(100)晶面,代表AC的石墨化程度[19],石墨化程度越高表明活性炭对非极性VOCs的吸附能力越强。AC/MgO-1%的衍射峰向较低的XRD角方向轻微移动。根据布拉格定律(2dsinθ=λ),可以看出由掺杂导致AC的晶格裕度(d)增加[21]。相较于AC,AC/MgO-0%在25°处的衍射峰强度增加不明显,AC/MgO-1%在25°和43°处的衍射峰强度增加明显。这说明负载MgO有利于AC石墨化程度加强。此外,AC/MgO-1%在42°和62°处出现衍射峰,说明在活性炭中的氯化镁经高温焙烧后,暴露出(200)晶面和(220)晶面,镁主要以MgO的形式存在AC中。
2) SEM和EDS分析。AC和AC/MgO-1%进行扫描电镜观察,结果如图7所示。由图7可以看出,AC和AC/MgO-1%的表面纹理存在差异,AC的表面光滑,而AC/MgO-1%的表面粗糙,部分区域甚至出现孔坍塌。这是因为在金属离子转变成金属氧化物的高温过程中,活性炭孔道会不断扩大而相互贯穿,最终导致坍塌,部分孔结构消失[22]。掺杂的金属氧化物均匀的负载在活性炭上,这为吸附弱吸附质提供了大量的吸附位点。如图8所示,从左到右分别是AC/MgO-1%Mg、C、O的EDS表征结果。可以看出,Mg成功掺杂在AC中,表明AC/MgO制备成功,且Mg在AC表面分散程度高。
3) BET分析。对AC、AC/MgO-0%、AC/MgO-0.5%、AC/MgO-1%、AC/MgO-1.5%和AC/MgO-2%样品进行BET分析,结果如表2所示。由表2可以看出,AC/MgO相比于AC,比表面积和总孔体积下降,可能是金属氧化物在AC中堵塞造成[15]。微孔比表面积和微孔体积上升,可能是金属离子转变成金属氧化物的过程中具有扩孔效应[23]。在不同掺杂量的AC/MgO中,AC/MgO-1%的微孔比表面积和微孔体积最大,为450 m2·g−1和0.211 cm3·g−1,说明AC/MgO-1%具有更大的空间吸附VOCs,整体呈现先增加后减少的趋势,可能是随着掺杂比例的提高,金属离子占据微孔的影响比扩孔效应大,导致比表面积和总孔体积减少,这与韩智广等[24]的研究结果相似。
4) FTIR谱图分析。AC和AC/MgO-1%样品的FTIR谱图如图9所示。由图9可以看出,AC/MgO-1%相比于AC,在1 070、1 589和3 435 cm−1处的峰均有所增加,3 435 cm−1处的吸收峰是—OH的吸收峰,2 826 cm−1附近的吸收峰是对称的C—H拉伸振动形成的,1 589 cm−1附近的吸收峰是羧基中的C=O振动引起的,1 385 cm−1附近的吸收峰是—OH的吸收峰,1 070 cm−1附近的吸收峰是C—O或C—H伸缩振动引起的,774 cm−1附近的吸收峰是—CH,568 cm−1附近的吸收峰是芳环中C—H的平面外弯曲振动引起。相比于AC,AC/MgO-1%中增加了对极性VOCs分子的吸附位点(C—O、—OH),进而提高了对极性VOCs分子的吸附容量[25]。
-
对活性炭模型和氧化镁晶胞优化结构如图10所示。活性炭模型通常采用石墨烯模型,用氢原子将周围活性位点占据[26]。本研究采用石墨烯模型且尽可能让吸附质完全覆盖,具有大多数活性炭的代表性。氧化镁模型采用氧化镁晶胞,氧化镁(220)晶面为吸附表面,氧化镁晶胞参数为a、b、c均为0.421 7 nm,α=90°、β=90°、γ=90°。
对活性炭和氧化镁吸附甲苯、乙酸乙酯、异丙醇模型的结构优化如图11(a)、(b)所示。表3中的吸附能通过式(2)计算得到。AC对甲苯、乙酸乙酯、异丙醇的吸附能分别为−21.98、−7.37、−3.09 kJ·mol−1,键长分别为0.35、0.361、0.275 nm;而MgO对甲苯、乙酸乙酯、异丙醇的吸附能分别为−6.21、−12.81、−23.70 kJ·mol−1,键长分别为0.29、0.25、0.22 nm。结果表明,甲苯更容易在AC上吸附,是由于甲苯中的离域π电子和AC中富电子相互作用导致其吸附能大[27]。异丙醇在MgO上的吸附能比甲苯大,可能是异丙醇中含有羟基官能团,导致其与MgO亲和力强于甲苯。因此,在AC中掺杂MgO可以提升对乙酸乙酯和异丙醇的吸附能力。
-
1)掺杂比例为1%、焙烧温度为450 ℃的AC/MgO对甲苯、乙酸乙酯、异丙醇的吸附性能最好,与AC相比,单组分时吸附甲苯、乙酸乙酯、异丙醇的吸附容量分别为326 mg·g−1、175 mg·g−1、124 mg·g−1,分别提高42%、18%、25%,在二元吸附时,弱吸附质在AC/MgO-1%的被取代量从51%、55%、38%下降至24%、44%、33%,吸附容量提高34%~80%,且经5次吸脱附循环实验后吸附容量仍维持在92.68%以上。
2)浸渍法制备的氧化镁掺杂活性炭,MgO可均匀负载在AC表面和内部,比表面积和总孔体积虽有所下降,但AC/MgO-1%的微孔表面积和微孔体积提高,含氧官能团和酸性官能团提高,对极性VOCs分子的吸附位点增加,吸附容量提高。
3)甲苯、乙酸乙酯及异丙醇在AC上吸附时甲苯为强吸附质,在氧化镁上吸附时异丙醇为强吸附质,在AC中掺杂氧化镁可提高吸附容量,同时可减小甲苯、乙酸乙酯及异丙醇的竞争吸附作用。
氧化镁掺杂改性活性炭的制备及其对彩板印刷VOCs废气的吸附性能及机理
Preparation of magnesium oxide doped modified activated carbon and its adsorption properties and mechanism toward color plates printing VOCs
-
摘要: 针对实际工业应用中活性炭吸附VOCs存在的竞争吸附导致吸附量减少的问题,以彩板印刷过程产生的甲苯、乙酸乙酯、异丙醇为代表,采用浸渍法对煤基活性炭进行掺杂氧化镁改性,提高活性炭在实际应用中多组分吸附体系的吸附容量。结果表明,氧化镁的最佳掺杂比和焙烧温度分别为1%和450 ℃,相比原始活性炭,AC/MgO-1%在单组分吸附实验中对甲苯、乙酸乙酯、异丙醇的吸附容量分别提高了42%、18%、25%,在二元吸附实验中,弱吸附质在AC/MgO-1%的被取代量由51%、55%、38%下降至24%、44%、33%,二元竞争作用减弱,吸附容量提高34%~80%;经五次吸脱附循环实验后,AC/MgO-1%的吸附容量仍维持在92.68%以上。DFT计算结果表明,甲苯、乙酸乙酯、异丙醇在AC上吸附的过程中,甲苯为强吸附质,在氧化镁上吸附时异丙醇为强吸附质,氧化镁掺杂可在一定程度上减少二元竞争作用;SEM、BET、FTIR及XRD表征结果表明,掺杂氧化镁虽会减少比表面积,但会增加其极性官能团和活性位点,提高吸附容量。Abstract: Aiming at the adsorption capacity reduction of VOCs on activated carbon due to the competitive adsorption in industrial applications, coal-based activated carbon was modified with magnesium oxide doped by impregnation to improve the adsorption capacity for multicomponent adsorption, taking toluene, ethyl acetate, and isopropanol as the representatives of VOCs in color plates printing. The results showed that the optimal doping ratio and calcination temperature of magnesium oxide were 1% and 450 ℃, respectively. Compared with the original activated carbon, the adsorption capacity of modified activated carbon to toluene, ethyl acetate and isopropanol in single-component adsorption experiments increased by 42%, 18% and 25%, respectively, the substitution amount of weak adsorbates in the binary adsorption experiments decreased from 51%, 55% and 38% to 24%, 44% and 33%, respectively, and the adsorption capacity increased by 34%~80%, indicating that the binary competition was weakened. The adsorption capacity of AC/MgO-1% remained above 92.68% after five regeneration cycles. DFT calculations showed that the adsorption process of toluene, ethyl acetate, and isopropanol, toluene was a type of strong adsorbate adsorbed on AC, while on magnesium oxide, isopropanol was a type of strong adsorbate, and magnesium oxide doping could reduce the binary competition effect to a certain extent. SEM, BET, FTIR, and XRD characterization results indicated that doping of magnesium oxide decreased the specific surface area, but it increased the polar functional groups and active sites, and improved the adsorption capacity.
-
Key words:
- activated carbon /
- modified /
- adsorption /
- VOCs /
- DFT
-
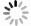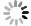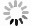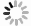
-
表 1 VOCs的物性参数
Table 1. Physical parameters of VOCs
VOCs 分子式 摩尔质量/(g·mol−1) 密度/(g·cm−3) 沸点/℃ 极性指数 动力学直径/nm 甲苯 C7H8 92.14 0.867 110.6 2.4 0.67 乙酸乙酯 C4H8O2 88.11 0.902 77.06 4.3 0.52 异丙醇 C3H8O 60.06 0.785 82.45 4.3 0.47 表 2 AC和AC/MgO的结构参数
Table 2. Structure parameters of AC and AC/MgO
样品 比表面积/
(m2·g−1)微孔比表面积/
(m2·g−1)介孔比表面积/
(m2·g−1)总孔体积/
(cm3·g−1)微孔体积/
(cm3·g−1)介孔体积/
(cm3·g−1)AC 763 433 94 0.614 0.205 0.310 AC/MgO-0 620 375 53 0.591 0.17 0.336 AC/MgO-0.5% 563 382 59 0.553 0.180 0.331 AC/MgO-1% 693 450 71 0.556 0.211 0.268 AC/MgO-1.5% 607 376 63 0.508 0.176 0.262 AC/MgO-2% 528 363 48 0.519 0.171 0.298 表 3 VOCs在活性炭和氧化镁上的吸附能和吸附键长
Table 3. Adsorption energy and bond length of VOCs on activated carbon and magnesium oxide
吸附分子 活性炭模型
吸附能/
(kJ·mol−1)活性炭模型
吸附键长/
nmMgO模型
吸附能/
(kJ·mol−1)MgO模型
吸附键长/
nm甲苯 −21.98 0.350 −6.21 0.29 乙酸乙酯 −7.37 0.316 −12.81 0.25 异丙醇 −3.09 0.275 −23.70 0.22 -
[1] MIAO G, WANG Y, WANG B, et al. Multi-omics analysis reveals hepatic lipid metabolism profiles and serum lipid biomarkers upon indoor relevant VOC exposure[J]. Environment International, 2023, 180: 108221. doi: 10.1016/j.envint.2023.108221 [2] WANG Y, CUI Y, HE Q, et al. Significant impact of VOCs emission from coking and coal/biomass combustion on O3 and SOA formation in taiyuan, China[J]. Atmospheric Pollution Research, 2023, 14(2): 101671. doi: 10.1016/j.apr.2023.101671 [3] 黄沛荣, 朱波, 张月, 等. PM2.5与O3协同控制视角下深圳市工业VOCs源谱特征[J]. 中国环境科学, 2022, 42(8): 3473-3482. doi: 10.3969/j.issn.1000-6923.2022.08.001 [4] 胡经纬, 苏静雯, 刘思彤, 等. 有机胺改性树脂基球形活性炭对二氧化碳的吸附行为 [J]. 华东理工大学学报(自然科学版), 2024, 50 (2): 192-198. [5] 党小庆, 王琪, 曹利, 等. 吸附法净化工业VOCs的研究进展[J]. 环境工程学报, 2021, 15(11): 3479-3492. doi: 10.12030/j.cjee.202011052 [6] 刘霞, 党小庆, 曹利, 等. NaY分子筛疏水改性及其对汽车涂装VOCs废气的吸附[J]. 环境工程学报, 2023, 17(11): 3689-3697. doi: 10.12030/j.cjee.202306092 [7] SHARMA H, DHIR A. Capture of carbon dioxide using solid carbonaceous and non-carbonaceous adsorbents: a review[J]. Environmental Chemistry Letters, 2021, 19(2): 851-873. doi: 10.1007/s10311-020-01118-2 [8] HUANG X, TANG M, LI H, et al. Adsorption of multicomponent VOCs on various biomass-derived hierarchical porous carbon: A study on adsorption mechanism and competitive effect[J]. Chemosphere, 2023, 313: 137513. doi: 10.1016/j.chemosphere.2022.137513 [9] YAO X, LIU Y, LI T, et al. Adsorption behavior of multicomponent volatile organic compounds on a citric acid residue waste-based activated carbon: Experiment and molecular simulation[J]. Journal of Hazardous Materials, 2020, 392: 122323. doi: 10.1016/j.jhazmat.2020.122323 [10] 侯博, 陈思铭, 江波, 等. 活性炭吸附挥发性有机化合物的研究进展[J]. 安全与环境工程, 2021, 28(1): 197-208. [11] 杨晓娜, 任晓玲, 严孝清, 等. 活性炭对VOCs的吸附研究进展[J]. 材料导报, 2021, 35(17): 17111-17124. doi: 10.11896/cldb.21010141 [12] RONG X, CAO Q, GAO Y, et al. Performance optimization and kinetic analysis of HNO3 coupled with microwave rapidly modified coconut shell activated carbon for VOCs adsorption[J]. Frontiers in Energy Research, 2023, 10: 1047254. doi: 10.3389/fenrg.2022.1047254 [13] BU N, LIU X, LI T, et al. Oxalic acid–modified activated carbons under hydrothermal condition for the adsorption of the 2-butanone[J]. Environmental Science and Pollution Research, 2023, 30(50): 109606-109617. doi: 10.1007/s11356-023-30095-6 [14] KANG Y J, KIM Y J, YOON S J, et al. Effective removal of acetaldehyde using piperazine/nitric acid co-impregnated bead-type activated carbon[J]. Membranes, 2023, 13(6): 595. doi: 10.3390/membranes13060595 [15] ZHOU K, MA W, ZENG Z, et al. Experimental and DFT study on the adsorption of VOCs on activated carbon/metal oxides composites[J]. Chemical Engineering Journal, 2019, 372: 1122-1133. doi: 10.1016/j.cej.2019.04.218 [16] 金春江, 王鲁元, 陈惠敏, 等. 一步快速活化法制备生物质活性炭及其对乙酸乙酯的吸附再生[J]. 化工进展, 2021, 40(S1): 446-455. [17] YANG F, LI W, ZHONG X, et al. The alkaline sites integrated into biomass-carbon reinforce selective adsorption of acetic acid: In situ implanting MgO during activation operation[J]. Separation and Purification Technology, 2022, 297: 121415. doi: 10.1016/j.seppur.2022.121415 [18] BAUR G B, YURANOV I, KIWI-MINSKER L. Activated carbon fibers modified by metal oxide as effective structured adsorbents for acetaldehyde[J]. Catalysis Today, 2015, 249: 252-258. doi: 10.1016/j.cattod.2014.11.021 [19] CHENG T, LI J, MA X, et al. Competitive adsorption characteristics of VOCs and water vapor by activated carbon prepared from Fe/N-doped pistachio shell[J]. Environmental Science and Pollution Research, 2023, 30(39): 91262-91275. doi: 10.1007/s11356-023-28509-6 [20] PELEKANI C, SNOEYINK V L. Competitive adsorption in natural water: role of activated carbon pore size[J]. Water Research, 1999, 33(5): 1209-1219. doi: 10.1016/S0043-1354(98)00329-7 [21] LIU Q, YANG F, LIU Z, et al. Preparation of SnO2–Co3O4/C biochar catalyst as a Lewis acid for corncob hydrolysis into furfural in water medium[J]. Journal of Industrial and Engineering Chemistry, 2015, 26: 46-54. doi: 10.1016/j.jiec.2014.11.041 [22] 党小庆, 敬开锐, 马红周, 等. 吸附VOCs活性炭真空热再生及影响因素实验[J]. 环境工程, 2022, 40(8): 62-68. [23] 徐州, 李伟, 刘守新. 载铜活性炭的制备及其气相苯吸附性能的研究[J]. 林产化学与工业, 2022, 42(3): 1-9. doi: 10.3969/j.issn.0253-2417.2022.03.001 [24] 韩智广, 刘寒冰, 陈传胜, 等. 碱铜联合改性珠状活性炭及其对甲苯的吸附机理[J]. 环境科学研究, 2021, 34(11): 2665-2673. [25] 黄钰坪, 王登辉, 惠世恩, 等. 生物炭材料吸附VOCs研究进展[J]. 洁净煤技术, 2022, 28(2): 40-53. [26] WANG H, GAO J, XU X, et al. Adsorption of volatile organic compounds (VOCs) on oxygen‐rich porous carbon materials obtained from glucose/potassium oxalate[J]. Chemistry–An Asian Journal, 2021, 16(9): 1118-1129. doi: 10.1002/asia.202100098 [27] 王正, 毛以朝, 杨清河, 等. 改性活性炭吸附芳烃的研究进展[J]. 应用化工, 2023, 52(12): 3406-3410. doi: 10.3969/j.issn.1671-3206.2023.12.037 -



 下载:
下载: