-
总悬浮颗粒(TSP)是粒径<100 μm的气溶胶粒子的总称. 大气中的气溶胶能吸收和散射太阳辐射,直接影响地表辐射强度和温度,也可以作为云凝结核或冰核,间接影响全球气候[1 − 3]. 此外细颗粒气溶胶(粒径小于2.5 μm)还能进入人体肺部,影响人体呼吸道与心血管健康[4 − 5].
海洋覆盖了地球71%的表面,海洋气溶胶长距离传输和沉降在海洋生源要素的循环中扮演重要的角色[6]. 海洋气溶胶来源包括一次来源(例如海盐和矿物等)和二次生成的无机和有机组分[7]. 其中,海盐是海洋大气气溶胶中最主要的自然源,全球每年大约有 3300 Tg海盐颗粒物通过风和波浪的作用进入大气[8]. 矿物颗粒物主要来源陆地传输,且研究表明东亚地区每年向大气传输约31—96 Tg沙尘,其中近 50%传输至太平洋[9]. 二次气溶胶主要是人为及自然源释放到大气中的气体(NOx、SO2和NH3等)在大气中通过一系列大气化学反应生成二次气溶胶(SO42−、NO3−和NH4+等)[10]. 陆地和人为活动释放到大气中的物质通过季风输送至我国近海及开阔大洋的大气[11],并通过干湿沉降到上层海洋,引起了海水酸化[12]. 无机氮(NO3−和NH4+)沉降到上层海洋后直接改变表层海水的N和P的比值,进而影响海洋初级生产力与全球氮循环[13 − 15].
对于西太平洋地区气溶胶的研究主要集中在西北太平洋及其边缘海(南海、黄海、东海和日本海等)[16 − 20],对赤道西太平洋的研究较少. 本研究以热带西太平洋为研究区域,通过航次采集了TSP样本,分析了TSP中主要水溶性离子的浓度,探讨了各离子的潜在来源以及估算西太平洋无机氮干沉降通量.
-
2015年12月5日—2016年1月10日,通过搭载科学考察船,利用大流量采样器采集了大气总悬浮颗粒物(TSP)样品29个. 采样点位置采样航迹如图1所示. 航迹始于中国厦门,途经菲律宾、印度尼西亚与巴布亚新几内亚附近海域,最终结束于中国黄海. TSP样品用KC-1000采样器采集,采样器固定在船体顶部甲板,与海平面距离约10 m,采样流量设为1.05 m3 min−1. TSP样品用PALL TissuquartzTM滤膜,采样前滤膜在马弗炉425 ℃烧4 h. TSP样本采集后至于洗净的自封袋中,冰箱−20 ℃保存. 采样过程中,设置2个空白滤膜,与样品滤膜在相同条件下进行保存,作为本航次的空白样品.
-
样品在锡箔纸上用陶瓷剪刀裁剪1/4张采样滤膜置于50 mL离心管中,加30 mL Mill-Q水(RephiLe Bioscience公司),水浴超声振荡萃取20 min,静置20 min,重复2次,用0.22 μm微孔滤膜过滤,空白滤膜采取同样的处理操作.
采用DionexIonPacTM AS22型离子色谱(Thermo scientific公司)Na2CO3溶液做淋洗液以1.2 mL·min−1速率测定溶液中的阴离子,用DionexIonPacTMCS12A型离子色谱甲基磺酸溶液做淋洗液以1.0 mL·min−1速率测定溶液中的阳离子. NO3−、SO42−、K+、Na+、Ca2+、Mg2+、NH4+离子的检出限分别为21.6、11.5、1.77、0.001、0.09、2.47、1.21 μg·L−1.
使用SIGMA-ALDRICH公司的阴阳离子标准液分别定标,后分析气溶胶样品的阴、阳离子(NO3−、SO42−、K+、Na+、Ca2+、Mg2+、NH4+)的浓度,为保证数据准确可靠,在测定过程中插入质控样,经计算本次实验相对标准偏差小于5%.
为了分析样本水溶性离子的化学计量学关系,用公式(1)气溶胶水溶性离子的空气质量浓度C(μg·m−3)转换为电荷浓度Ce(neq·m−3):
式(1)中,C为某离子的空气质量浓度;Ce为某离子的电荷浓度;n为某离子的正或负整数电荷;M为某离子的相对分子质量. 样本中海洋源离子的电荷浓度与Na+的电荷浓度的关系由式(2)—(5)计算:
式中,[ss-Mg2+]、[ss-K+]、[ss-Ca2+]和[ss-SO42−]是电荷浓度(neq·m−3)时,系数a、b、c和d分别为0.23、0.022、0.044和0.121[21].
再用SPSS25.0软件对样本中水溶性离子的浓度进行相关性分析和主成分旋转因子载荷矩阵分析.
无机氮干沉降通量的计算公式如下:
式中,C是气溶胶物种的大气浓度,V是干沉降速度. NO3−、NH4+的干沉降速率的空间变化率主要受颗粒物粒径、风速、相对湿度和海面粗糙度等因素影响[22 − 23],根据前人的研究,干沉积速度变化超过3个数量级[23]. NO3−与NH4+粒径主要分布在10 µm以内[24 − 25]. 根据无机氮沉降模型和实验结果[26 − 27]以及上述NH4+、NO3−在颗粒中的尺寸分布,NO3−的沉降速度为0.72 cm·s−1,NH4+的沉降速率为0.77 cm·s−1.
-
为确定中国近海及西太平洋气溶胶的可能来源,使用美国国家海洋与大气管理局(NOAA)大气资源实验室(ARL)提供的气象数据(https://ready.arl.noaa.gov/)和混合单粒子拉格朗日积分轨道(HYSPLIT)模型(https://www.ready.noaa.gov/documents/Tutorial/html/index.html)在经纬度1° × 1°分辨率内对气团后向轨迹进行分析. 气团后向轨迹回追起始时间为采样开始的时刻,轨迹运行的起始点海拔高度为800 m,气团后向轨迹分析时间总计为120 h.
-
为研究西太平洋地区海洋气溶胶中主要水溶性离子的浓度水平及化学组成的差异,引入3组他人研究中的观测数据与本次研究对比,一为Li等[28]所研究的彭佳屿的TSP样本数据,二为Wang等[29]所研究的太平洋西边界的TSP样本数据,三为Kunwar 等[30]所研究的冲绳岛数据,其航线与采样点如图1所示.
-
基于2015年冬季气团后向轨迹得到采集期间采样点的气团输入来源情况(图1). 本次研究大部分位点的气团直接来源于开阔大洋但1、2、17、28与29号位点的气团来源复杂,1号气团经过台湾岛北部,2号位点经过菲律宾北部,17号位点来源于东南亚岛屿地区,28和29号位点来源于蒙古国与俄罗斯北部地区.
故依据采样点是否受陆源气团影响将1、2、17、28、29号位点的样本归类为受到陆源气团影响的近海海域样本,其余作为受海洋气团影响的开阔大洋样本.
-
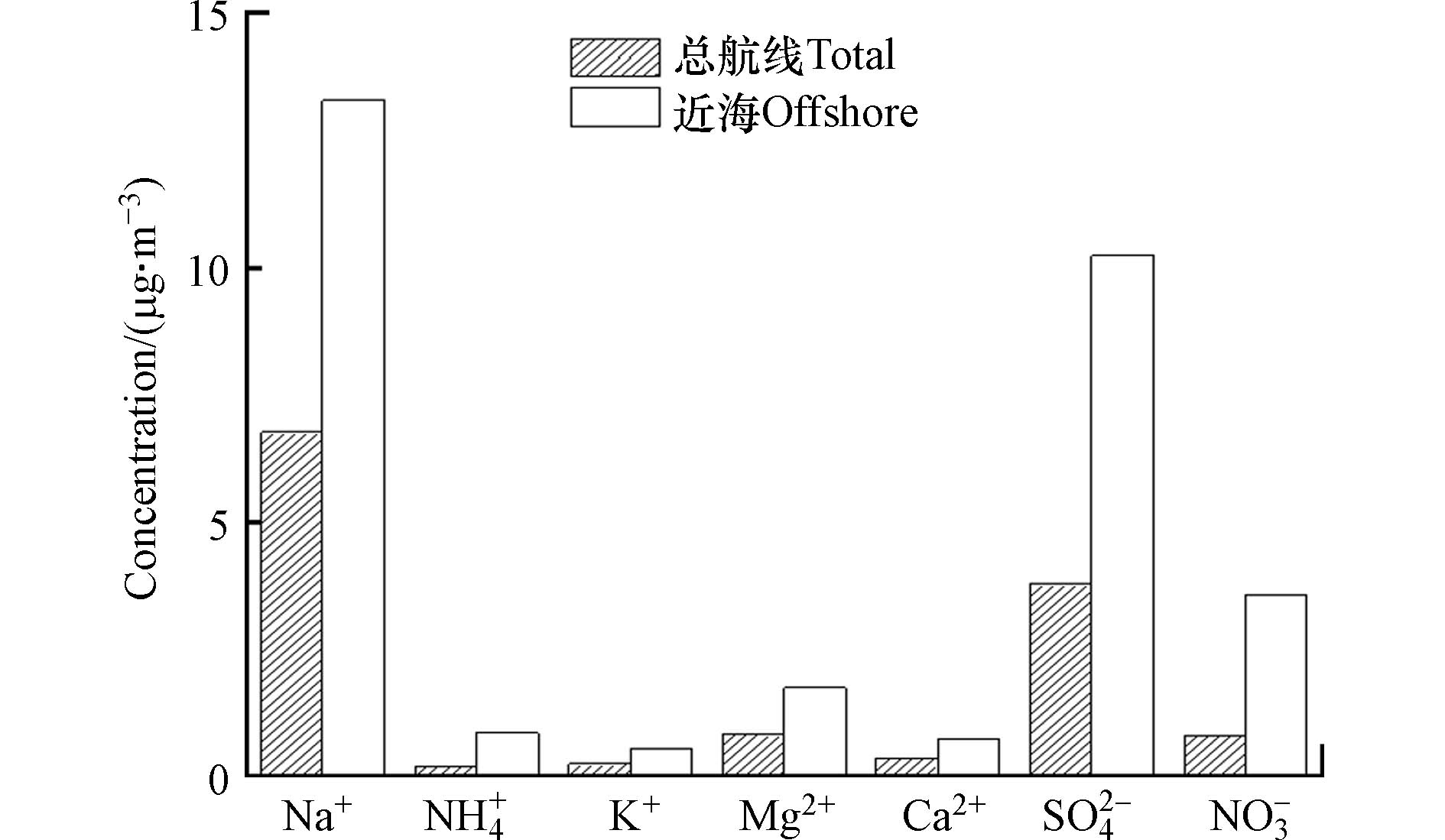
本次实验所测得的7种水溶性离子浓度如表1所示,所测定的离子在不同时空有很大的变化,反映了大气化学成分及来源贡献的巨大空间差异. 7种离子的浓度排序为Na+ > SO42− > Mg2+ > NO3− > Ca2+ > K+ > NH4+,阴离子中SO42−浓度最高,平均为(3.78 ± 3.81)µg·m−3,占总离子质量浓度的29.1%,阳离子中Na+浓度最高,平均为(6.76 ± 5.92)µg m−3,占总离子质量浓度的52.0%. Na+的占比说明海盐对海面上气溶胶的化学组成有着重要的贡献[31].
图2将总体样本与近海样本中各离子平均质量浓度作比较,可以清晰看出受陆源影响的气溶胶样本各离子浓度均高于整体平均值,其中Na+、NH4+、NO3−与SO42−最为显著,经计算得近海海域贡献了本航次74.9%的无机氮(NO3−、NH4+)与46.6% SO42−. NO3−由前体物NOx光化学氧化形成,NOx主要来源于生物质燃烧、煤燃烧、机动车尾气及土壤微生物排放;NH4+由NH3转化而来,NH3的来源较复杂包括燃料燃烧、农业活动等[32 − 33]. 此外,近海样本中的地壳离子(Mg2+、Ca2+)受陆源气团的影响浓度高于总体均值.
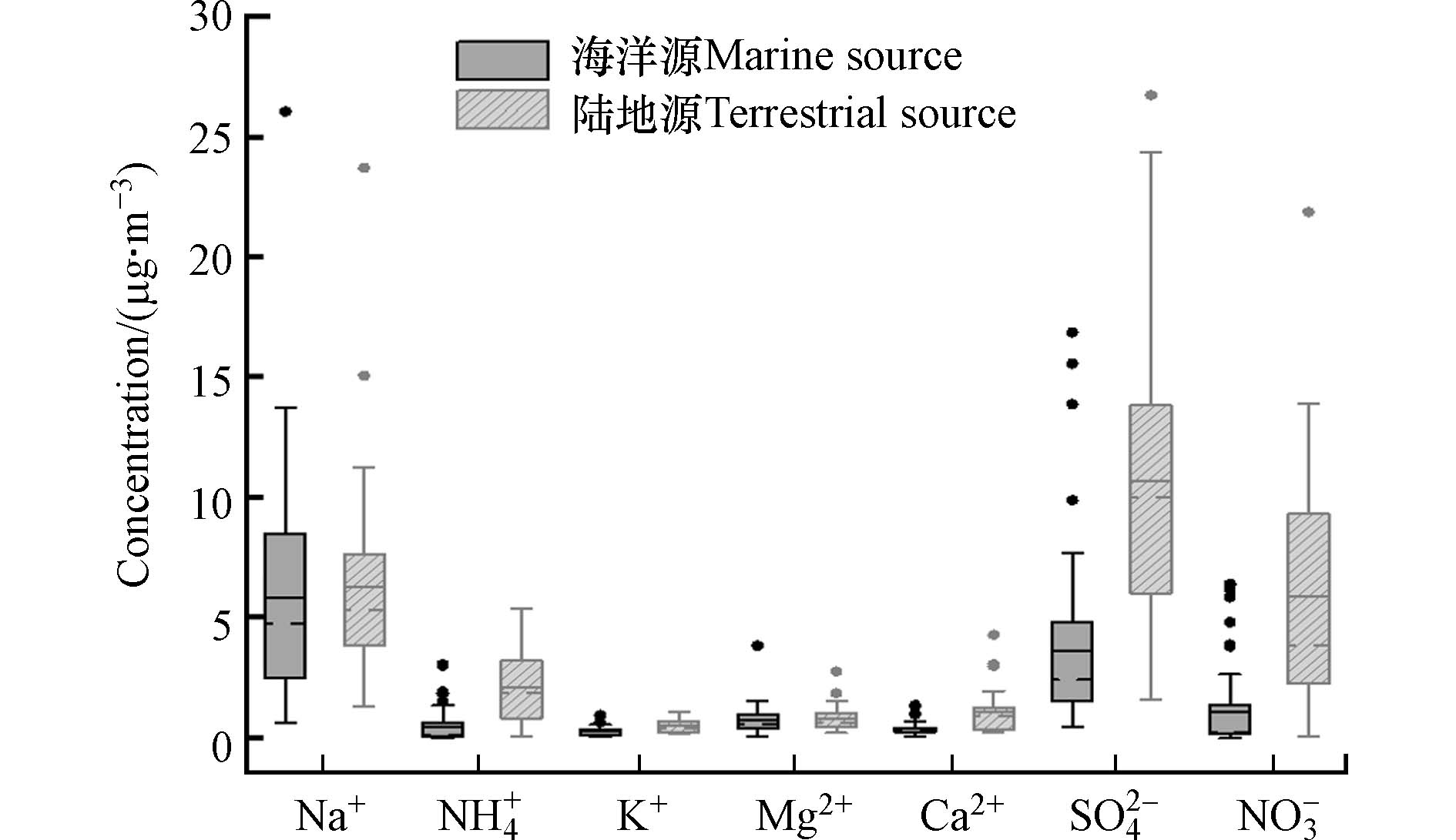
为厘清亚洲东部陆缘海及开阔大洋气溶胶水溶性离子的空间分布特征,汇编了彭佳屿[28]、太平洋西边界航次[29]、冲绳岛[30]、本航次共四地7种水溶性离子的数据(图3). Na+、K+、Mg2+以海洋源为主,浓度差异小. 彭佳屿与冲绳岛的Ca2+离子浓度高于本研究,推测这是因为岛屿地区受到地壳矿物的影响[11];NH4+、NO3−和SO42− 等3种离子以彭佳屿的浓度最高,说明冬季彭佳屿受大陆气团影响最大. 图4对比表明主要受海洋源气团和陆地源气团影响的样本中各离子浓度的差异,受陆地源气团的样本中NH4+、NO3−和SO42− 等3类典型的二次离子与地壳离子Ca2+的浓度远高于海洋源的气团. 因此,对比讨论受陆源气团影响严重的近海海域和主要受海洋气团影响的开阔大洋地区TSP中离子的化学成分十分必要.
-
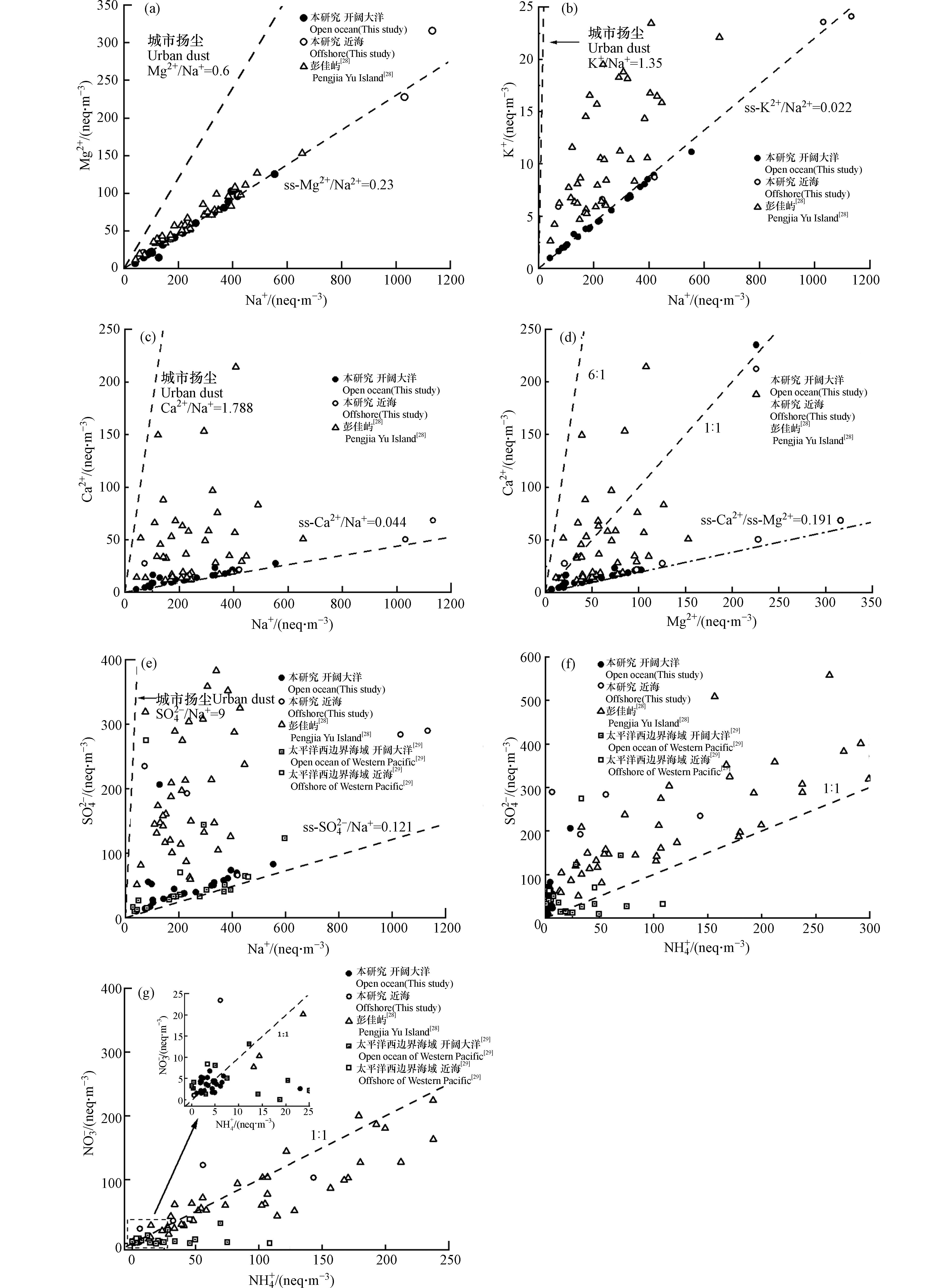
Na+是海洋气溶胶中最主要的水溶性离子,且主要来自海洋源,不会因发生置换反应而亏损,故使用Na+作为参考离子讨论海水对海洋气溶胶中其他水溶性离子的贡献[34]. 本研究样本中Mg2+/Na+均在0.23附近,即开阔大洋中Mg2+来源基本均来自于海盐;近海海域的样本浓度较高但比值仍在0.23附近,说明Mg2+主要来源于海盐. 彭佳屿的Mg2+/Na+高于0.23,说明Mg2+大部分来自于海洋,但受到土壤地壳的影响(图5a). 本研究K+的情况与Mg2+相似,K+/Na+在0.022附近,说明中近海与开阔大洋的K+的主要来源是海盐. 但彭佳屿的离散程度较大,比值均高于0.22但小于城市扬尘的K+/Na+=1.35,这说明彭佳屿K+可能受到陆源的影响,其可能来自陆源矿物和土壤,以及生物质燃烧和煤燃烧释放等[35](图5b). 本研究全部样品的Ca2+/Na+均略高于0.044(图5c),说明Ca2+受到陆源的影响. 彭佳屿的Ca2+浓度显著较高,且Ca2+/Na+值大于0.044,说明有大量nss-Ca2+存在,nss-Ca2+主要来自地壳和当地建筑粉尘. 建筑粉尘中Ca/Mg为10—82,远高于地壳矿物中Ca/Mg(1—6)[36],而本航次样品Ca2+/Mg2+几乎均小于1(图5d),这进一步证明了本航次Ca2+主要为海洋来源,仅有少量地壳来源. 本研究与Wang等[29]的研究所有样本的SO42−/Na+均大于0.121(图5e),说明除海盐外,海洋SO42−气溶胶存在外源贡献,在开阔大洋区域这可能是由于表层海水浮游植物在代谢活动中会向大气释放二甲基硫(DMS),二甲基硫在大气中被氧化成为甲基磺酸盐(MSA),或被最终氧化为nss-SO42-[37]. 值得注意的是两者的近海海域及彭佳屿样品的SO42−/Na+均远大于0.121,说明冬季大陆气团是近海气溶胶SO42−的主要贡献者.
SO42−的形成与SO2的排放密不可分. 人为SO2的排放,即以煤为主的含硫物质燃烧释放的SO2在大气中通过气相、液相或光化学等氧化反应最终生成SO42−,由东亚季风输送至近海上空. 气溶胶中的SO42−、NO3−、NH4+大多以(NH4)2SO4或NH4HSO4和NH4NO3的形式存在[38]. 气溶胶样本中NH4+在中和SO42−、NO3−的方面有重要作用但3组样品的绝大多数样本的SO42−/NH4+均大于1:1,说明NH4+不能完全中和SO42−(图5f ). 彭佳屿与太平洋西边界的样本中存在少量样品NO3−/NH4+小于1:1,说明其样本中有NH4NO3存在(图5g).
气溶胶中水溶性离子的相关性分析与主成分分析可以反映两离子间的密切程度,可能来源与共存方式,在指示其相似的产生、运输与移除机制中起着重要作用. 两者的结果共同显示Na+、Ca2+、K+和Mg2+主要为海洋源;而NH4+、NO3−主要为陆地源;SO42−及存在海洋又存在陆地来源. 表2所示开阔大洋和近海海域Na+、Ca2+、K+、Mg2+离子两两间的相关性系数均大于0.893且存在显著的相关性(P < 0.05). 说明该组的Na+、Ca2+、K+、Mg2+4种离子主要来源于海洋源. 此外开阔大洋样本中NH4+与SO42−也存在显著相关性,说明样本中有(NH4)2SO4存在. 表3所示开阔大洋样本2个主成分因子解释了84.2%的来源,其特征值均大于1. Na+、Ca2+、K+、Mg2+等海盐离子在成分1中的高载荷说明因子1可能是海洋源,因子1解释了57.7%的变量,对Na+、K+、Mg2+和Ca2+4者有非常高的载荷分别为0.988、0.992、0.980和0.945. 因其均是海洋中常见的离子所以因子1代表海洋源. 因子2解释了26.5%的变量,其与NH4+和SO42−的载荷为0.985和0.904,说明因子2是二次源. 同理,近海海域样本两个主成分因子解释了94.5%的来源,推测因子一为海洋源,因子二为二次源.
-
本研究采用Qi等[27]所模拟的NO3−的沉降速度为0.72 cm·s−1,NH4+的沉降速率为0.77 cm·s−1. 根据近海海域及开阔大洋的TSP中的NO3−和NH4+的浓度,由公式(6)算得本航次的氮沉降通量. 其中,开阔大洋的NO3−-N的沉降通量为(0.14 ± 0.60)mg·m−2·d−1,NH4+-N的沉降通量为 (0.05 ± 0.05)mg·m−2·d−1. 近海海域的NO3−-N的沉降通量为(2.78 ± 2.61)mg·m−2·d−1,NH4+-N的沉降通量为(0.71 ± 0.71)mg·m−2·d−1. 近海海域的无机氮干沉降通量(3.47 ± 2.44)mg·m−2·d−1远大于开阔大洋的无机氮干沉降通量(0.20 ± 0.07)mg·m−2·d−1. 假设由大气无机氮沉降输入的营养盐全部可以被真光层的浮游植物吸收,由Redfield比值大致可估算出西太平洋开阔大洋大气无机氮沉降增加的初级生产力为(0.15 ± 0.08)g·m−2·a−1,近海海域初级生产力为(1.96 ± 1.95)g·m−2·a−1. 但由于海水表层营养盐结构也对浮游植物的吸收产生影响[39],所以上述数据为大气无机氮沉降增加初级生产力的理论最大值.
根据Qi等[27]研究中的各地冬季气溶胶无机氮浓度,根据上述公式计算出各地冬季无机氮沉降通量并进行横向对比(表4),可见中国近海至西太平洋N沉降通量的显著的空间差异,黄渤海的无机氮沉降通量最大(7.26 ± 3.83)mg·m−2·d−1,本航次开阔大洋的无机氮沉降通量最小(0.20 ± 0.17)mg·m−2·d−1. 但总体趋势是越靠南且远离大陆的地区无机氮沉降通量越小;纬度相近的开阔海洋比岛屿地区的无机氮沉降通量小.
-
本研究首先对冬季西太平洋29个TSP样本行后向轨迹分析,以受陆源或海源气团的影响分为近海海域样本与开阔大洋样本、再对两组水溶性离子进行成分分析与源解析并且计算了无机氮沉降通量与初级生产力的增加量.
结果显示,7种水溶性离子的总浓度的顺序为:Na+ > SO42− > Mg2+ > NO3− > Ca2+ > K+ > NH4+,TSP样本中Na+、K+和Mg2+主要来源于海洋源,SO42−、NO3−和NH4+主要来源于气溶胶在大气中的二次反应,且SO42−、NO3−、NH4+ 和Ca2+浓度从近海到开阔大洋呈现明显的下降趋势. 表明人为排放源和地壳源对近海海洋气溶胶中NO3−、NH4+ 和Ca2+的影响较大. 无机氮沉降通量在近海(3.47 ± 2.44)mg·m−2·d−1明显高于开阔大洋(0.20 ± 0.07)mg·m−2·d−1. 假设由大气无机氮沉降输入的营养盐全部可以被真光层的浮游植物吸收,根据Redfield比值可估算出西太平洋初级生产力增加的理论最大值,开阔大洋为(0.15 ± 0.08)g·m−2·a−1,近海海域为(1.96 ± 1.95)g·m−2·a−1.
冬季西太平洋TSP水溶性离子空间分布特征、来源和氮沉降
Spatial distribution and origin of TSP water-soluble ions, and nitrogen dry deposition in the Western Pacific Ocean in winter
-
摘要: 2015年12月5日至2016年1月10日,通过航次在西太平洋(WPO)采集了29个大气总悬浮颗粒物(TSP)样本,分析了TSP样本中水溶性离子(Na+、K+、NH4+、Mg2+、Ca2+、NO3−、SO42−)的浓度. 结合气团后向轨迹、化学计量学关系和主成分分析,对TSP中水溶性离子来源进行了分析. 结果显示,水溶性离子的总浓度的顺序为:Na+ >SO42− >Mg2+ >NO3− >Ca2+ >K+ >NH4+,其中Na+质量浓度为 (6.76 ± 5.92) μg·m−3,占总离子的52.0%;SO42−质量浓度 (3.78 ± 3.81) μg·m−3,占总离子浓度29.1%. SO42−、NO3−、NH4+ 和Ca2+浓度从近海到开阔大洋呈现明显的下降趋势,表明人为排放源和地壳源对近海海洋气溶胶中NO3−、NH4+ 和Ca2+的影响较大. 无机氮沉降通量在近海 (3.47 ± 2.44) mg·m−2·d−1明显高于开阔大洋 (0.20 ± 0.07) mg·m−2·d−1.Abstract: A total of 29 total suspended particulates (TSP) were collected over the Western Pacific Ocean (WPO) from December 5, 2015 to January 10, 2016 by cruise. Concentrations of water-soluble inorganic ions (Na+、K+、NH4+、Mg2+、Ca2+、NO3−、SO42−) were analyzed. The sources of water-soluble inorganic ions were investigated by together with air masses backward trajectories, stoichiometric analysis and principal component analysis. Results showed that the order of concentrations of water-soluble inorganic ions are Na+ > SO42− > Mg2+ > NO3− > Ca2+ > K+ > NH4+. Na+ ((6.76 ± 5.92) μg·m−3) and SO42− ((3.78 ± 3.81) μg·m−3) were the main ions, accounted for 52.0% and 29.1% mass concentrations of water-soluble inorganic ions. Concentrations of SO42−、NO3−、NH4+ and Ca2+ displayed decreased patterns from the offshore to the open ocean, suggested that the contributions of anthropogenic emissions and crust to those ions. The dry deposition fluxes of NO3− and NH4+ in offshore ((3.47 ± 2.44) mg·m−2·d−1) were obviously higher than those in open ocean ((0.20 ± 0.07) mg·m−2·d−1).
-
Key words:
- western pacific ocean /
- TSP /
- water-soluble ions /
- source apportionment /
- nitrogen deposition.
-
-
表 1 西太平洋冬季大气气溶胶中水溶性离子的浓度(µg·m−3)
Table 1. Concentrations of water-soluble ions in the atmospheric aerosols of Western Pacific in winter (μg·m−3)
Na+/(μg·m−3) NH4+/(μg·m−3) K+/(μg·m−3) Mg2+/(μg·m−3) Ca2+/(μg·m−3) SO42−/(μg·m−3) NO3−/(μg·m−3) 最大值 26.1 2.58 0.94 3.84 1.37 13.9 7.64 最小值 0.95 0.01 0.04 0.08 0.06 0.46 0.07 均值±标准差 6.76±5.92 0.21±0.50 0.25±0.22 0.83±0.80 0.36±0.27 3.78±3.81 0.80±1.79 表 2 冬季西太平洋TSP中水溶性离子相关性分析
Table 2. Correlation analysis of spring TSP water-soluble ions
开阔大洋(n = 24)
The open seaNa+ NH4+ K+ Mg2+ Ca2+ SO42− NO3− Na+ 1 NH4+ −0.19 1 K+ 0.998** −0.158 1 Mg2+ 0.993** −0.264 0.991** 1 Ca2+ 0.909** 0.046 0.914** 0.893** 1 SO42− 0.286 0.834** 0.321 0.218 0.420* 1 NO3− 0.209 −0.074 0.219 0.239 0.135 0.058 1 近海海域(n = 5)
Offshore areasNa+ NH4+ K+ Mg2+ Ca2+ SO42− NO3− Na+ 1 NH4+ −0.496 1 K+ 0.987** −0.352 1 Mg2+ 0.984** −0.509 0.966** 1 Ca2+ 0.903* −0.205 0.933* 0.939* 1 SO42− 0.536 0.319 0.652 0.55 0.727 1 NO3− 0.039 0.762 0.188 −0.055 0.151 0.608 1 注:*和**表示相关性达到显著(P < 0.05)和极显著水平(P < 0.01).
Note: * and ** indicate significant (P<0.05) and extremely significant levels of correlation (P < 0.01).表 3 主成分旋转因子载荷矩阵
Table 3. Rotated component matrix of major components
开阔大洋(n = 24)
The open sea近海海域(n = 5)
Offshore areas离子Ions 因子1Factor 1 因子2Factor 2 因子1Factor 1 因子2Factor 2 Na+ 0.988 −0.102 0.976 −0.153 NH4+ −0.087 0.985 −0.35 0.91 K+ 0.992 −0.068 0.99 0.009 Mg2+ 0.98 −0.177 0.98 −0.194 Ca2+ 0.945 0.115 0.964 0.087 SO42− 0.38 0.904 0.691 0.632 NO3− 0.268 −0.097 0.137 0.936 方差贡献率 57.7% 26.5% 63.5% 31.0% 累计贡献率 57.7% 84.2% 63.5% 94.5% 可能来源 海洋源 二次源 海洋源 二次源 表 4 冬季各地及本研究的无机氮沉降通量比较
Table 4. Comparison of inorganic nitrogen deposition fluxes in different regions with this study during winter
DDF NO3−-N/(mg·m−2·d−1) NH4+-N/(mg·m−2·d−1) Inorganic N/(mg·m−2·d−1) 开阔大洋(本研究) 0.14±0.60 0.05±0.05 0.20±0.07 近海海域(本研究) 2.78±2.61 0.71±0.71 3.47±2.44 太平洋西边界航线[29] 0.95±2.93 0.36±0.35 1.29±2.97 彭佳屿[28] 3.38±2.31 1.38±1.03 4.75±3.26 冲绳岛[30] 1.31±0.58 0.54±0.27 1.84±0.85 南海[27] 0.83±0.84 0.64±0.42 1.47±1.26 东海[27] 3.78±3.06 0.97±0.62 4.75±3.67 黄渤海[27] 5.24±2.95 2.02±0.88 7.26±3.83 -
[1] BHUYAN P, BARMAN N, BORA J, et al. Attributes of aerosol bound water soluble ions and carbon, and their relationships with AOD over the Brahmaputra Valley[J]. Atmospheric Environment, 2016, 142: 194-209. doi: 10.1016/j.atmosenv.2016.07.045 [2] 邢建伟, 宋金明. 中国近海大气颗粒物来源解析研究进展[J]. 环境化学, 2023, 42(3): 942-962. XING J W, SONG J M. Source apportionment of atmospheric particulates in China sea: A review[J]. Environmental Chemistry, 2023, 42(3): 942-962(in Chinese).
[3] O'DOWD C D, FACCHINI M C, CAVALLI F, et al. Biogenically driven organic contribution to marine aerosol[J]. Nature, 2004, 431(7009): 676-680. doi: 10.1038/nature02959 [4] MA J X, QI X, CHEN H X, et al. Coronavirus disease 2019 patients in earlier stages exhaled millions of severe acute respiratory syndrome coronavirus 2 per hour[J]. Clinical Infectious Diseases, 2021, 72(10): e652-e654. doi: 10.1093/cid/ciaa1283 [5] POPE III C A, DOCKERY D W. Health effects of fine particulate air pollution: lines that connect[J]. Journal of the Air & Waste Management Association, 2006, 56(6): 709-742. [6] KIM I N, LEE K, GRUBER N, et al. Increasing anthropogenic nitrogen in the North Pacific Ocean[J]. Science, 2014, 346(6213): 1102-1106. doi: 10.1126/science.1258396 [7] ARMOUR K C, MARSHALL J, SCOTT J R, et al. Southern Ocean warming delayed by circumpolar upwelling and equatorward transport[J]. Nature Geoscience, 2016, 9(7): 549-554. doi: 10.1038/ngeo2731 [8] ROTH B, OKADA K. On the modification of sea-salt particles in the coastal atmosphere[J]. Atmospheric Environment, 1998, 32(9): 1555-1569. doi: 10.1016/S1352-2310(97)00378-6 [9] LAMARE M L, LEE-TAYLOR J, KING M D. The impact of atmospheric mineral aerosol deposition on the albedo of snow & sea ice: are snow and sea ice optical properties more important than mineral aerosol optical properties?[J]. Atmospheric Chemistry and Physics, 2016, 16(2): 843-860. doi: 10.5194/acp-16-843-2016 [10] 姚星灿, 康汉青, 杜亚宁, 等. 基于二次无机气溶胶研究南京冬春季霾污染过程的形成特征和来源解析[J]. 环境化学, 2023, 42(12): 4151-4160. YAO X C, KANG H Q, DU Y N, et al. Formation characteristics and source analysis of haze pollution process during winter and spring in Nanjing based on secondary inorganic aerosol[J]. Environmental Chemistry, 2023, 42(12): 4151-4160(in Chinese).
[11] 罗笠, 高树基, 肖化云, 等. 台湾彭佳屿岛春季 TSP 中水溶性离子源解析[J]. 中国环境科学, 2017, 37(12): 4452-4459. LUO L, GAO S J, XIAO H Y, et al. Source apportionment of water-soluble ions in spring TSP of Pengjia Islet, Taiwan[J]. China Environmental Science, 2017, 37(12): 4452-4459(in Chinese).
[12] RIGNOT E, JACOBS S, MOUGINOT J, et al. Ice-shelf melting around Antarctica[J]. Science, 2013, 341(6143): 266-270. doi: 10.1126/science.1235798 [13] KIM T W, LEE K, NAJJAR R G, et al. Increasing N abundance in the northwestern Pacific Ocean due to atmospheric nitrogen deposition[J]. Science, 2011, 334(6055): 505-509. doi: 10.1126/science.1206583 [14] BIKKINA P, SARMA V, KAWAMURA K, et al. Dry-deposition of inorganic and organic nitrogen aerosols to the Arabian Sea: sources, transport and biogeochemical significance in surface waters[J]. Marine Chemistry, 2021, 231: 103938. doi: 10.1016/j.marchem.2021.103938 [15] OKIN G S, BAKER A R, TEGEN I, et al. Impacts of atmospheric nutrient deposition on marine productivity: Roles of nitrogen, phosphorus, and iron[J]. Global Biogeochemical Cycles, 2011, 25(2). [16] PARK G H, LEE S E, KIM Y, et al. Atmospheric deposition of anthropogenic inorganic nitrogen in airborne particles and precipitation in the East Sea in the northwestern Pacific Ocean[J]. Science of the Total Environment, 2019, 681: 400-412. doi: 10.1016/j.scitotenv.2019.05.135 [17] 石金辉, 张云, 高会旺, 等. 东海大气气溶胶的化学特征及来源[J]. 环境科学学报, 2011, 31(8): 1750-1757. SHI J H, ZHANG Y, GAO H W, et al. Characteristics and sources of atmospheric aerosols over the East China Sea[J]. Acta Scientiae Circumstantiae, 2011, 31(8): 1750-1757(in Chinese).
[18] HSU S C, WONG G T F, GONG G C, et al. Sources, solubility, and dry deposition of aerosol trace elements over the East China Sea[J]. Marine Chemistry, 2010, 120(1-4): 116-127. doi: 10.1016/j.marchem.2008.10.003 [19] 丁晓君, 代威力, 龙爱民, 等. 南海气溶胶中溶解性无机磷的空间和季节分布特征[J]. 环境化学, 2022, 41(7): 2347-2355. DING X J, DAI W L, LONG A M, et al. Spatial and seasonal distribution characteristics of dissolved inorganic phosphate of atmospheric aerosols over the South China Sea[J]. Environmental Chemistry, 2022, 41(7): 2347-2355(in Chinese).
[20] CUI X D, PENG L Y, GUO Y, et al. Distribution, source identification and ecological effects of aerosol dissolved nutrients in the Bohai Bay[J]. Environmental Pollution, 2023, 320: 121069. doi: 10.1016/j.envpol.2023.121069 [21] 陈敏. 化学海洋学[M]. 北京: 海洋出版社, 2009. CHEN M. Chemical oceanography [M]. Beijing: Ocean Press, 2009(in Chinese).
[22] DUCE R A, LISS P S, MERRILL J T, et al. The atmospheric input of trace species to the world ocean[J]. Global biogeochemical cycles, 1991, 5(3): 193-259. doi: 10.1029/91GB01778 [23] HOPPEL W A, FRICK G M, FITZGERALD J W. Surface source function for sea‐salt aerosol and aerosol dry deposition to the ocean surface[J]. Journal of Geophysical Research: Atmospheres, 2002, 107(D19): AAC 7-1-AAC 7-17. [24] NAKAMURA T, MATSUMOTO K, UEMATSU M. Chemical characteristics of aerosols transported from Asia to the East China Sea: an evaluation of anthropogenic combined nitrogen deposition in autumn[J]. Atmospheric Environment, 2005, 39(9): 1749-1758. [25] BAKER A R, LESWORTH T, ADAMS C, et al. Estimation of atmospheric nutrient inputs to the Atlantic Ocean from 50 N to 50 S based on large‐scale field sampling: Fixed nitrogen and dry deposition of phosphorus[J]. Global Biogeochemical Cycles, 2010, 24(3): .3634 [26] BAKER A R, ADAMS C, BELL T G, et al. Estimation of atmospheric nutrient inputs to the Atlantic Ocean from 50° N to 50° S based on large‐scale field sampling: Iron and other dust‐associated elements[J]. Global Biogeochemical Cycles, 2013, 27(3): 755-767. doi: 10.1002/gbc.20062 [27] QI J H, YU Y, YAO X H, et al. Dry deposition fluxes of inorganic nitrogen and phosphorus in atmospheric aerosols over the Marginal Seas and Northwest Pacific[J]. Atmospheric research, 2020, 245: 105076. doi: 10.1016/j.atmosres.2020.105076 [28] LI Y X, LIU S H, LUO L, et al. Seasonal variations, source apportionment and dry deposition of chemical species of total suspended particulate in Pengjia Yu Island, East China Sea[J]. Marine Pollution Bulletin, 2023, 187: 114608. doi: 10.1016/j.marpolbul.2023.114608 [29] WANG Q D, SONG J M, LI X G, et al. Geochemical characteristics and potential biogeochemical effect of water-soluble ions in atmospheric aerosols over the western boundary regions of Pacific Ocean[J]. Atmospheric research, 2019, 227: 101-111. doi: 10.1016/j.atmosres.2019.04.024 [30] KUNWAR B, KAWAMURA K. One-year observations of carbonaceous and nitrogenous components and major ions in the aerosols from subtropical Okinawa Island, an outflow region of Asian dusts[J]. Atmospheric Chemistry and Physics, 2014, 14(4): 1819-1836. doi: 10.5194/acp-14-1819-2014 [31] JIANG B, XIE Z, LAM P K S, et al. Spatial and temporal distribution of sea salt aerosol mass concentrations in the marine boundary layer from the Arctic to the Antarctic[J]. Journal of Geophysical Research:Atmospheres, 2021, 126(6): e2020JD033892. doi: 10.1029/2020JD033892 [32] LUO L, YAO X H, GAO H W, et al. Nitrogen speciation in various types of aerosols in spring over the northwestern Pacific Ocean[J]. Atmospheric Chemistry and Physics, 2016, 16(1): 325-341. doi: 10.5194/acp-16-325-2016 [33] SHI Y, XIA Y, LU B, et al. Emission inventory and trends of NO x for China, 2000–2020[J]. Journal of Zhejiang University Science A, 2014, 15: 454-464. doi: 10.1631/jzus.A1300379 [34] YEATMAN S G, SPOKES L J, JICKELLS T D. Comparisons of coarse-mode aerosol nitrate and ammonium at two polluted coastal sites[J]. Atmospheric Environment, 2001, 35(7): 1321-1335. doi: 10.1016/S1352-2310(00)00452-0 [35] SAVOIE D L, PROSPERO J M. Water‐soluble potassium, calcium, and magnesium in the aerosols over the tropical North Atlantic[J]. Journal of Geophysical Research:Oceans, 1980, 85(C1): 385-392. doi: 10.1029/JC085iC01p00385 [36] 黄辉军, 刘红年, 蒋维楣, 等. 南京市PM2.5物理化学特性及来源解析[J]. 气候与环境研究, 2006, 11(6): 713-722. HUANG H J, LIU H N, JIANG W M, et al. Physical and chemical characteristics and source apportionment of PM2.5 in Nanjing[J]. Climatic and Environmental Research, 2006, 11(6): 713-722 (in Chinese).
[37] REMPILLO O T, SEGUIN A, NORMAN A L. Shipboard measurement of atmospheric DMS and its oxidation products over the Canadian Arctic [C]//AGU Fall Meeting Abstracts. 2009, 2009: A43A-0184. [38] WANG Y, ZHUANG G S, ZHANG X Y, et al. The ion chemistry, seasonal cycle, and sources of PM2. 5 and TSP aerosol in Shanghai[J]. Atmospheric Environment, 2006, 40(16): 2935-2952. [39] 邢建伟, 宋金明, 袁华茂, 等. 胶州湾生源要素的大气沉降及其生态效应研究进展[J]. 应用生态学报, 2017, 28(1): 353-366. XING J W, SONG J M, YUAN H M, et al. Atmospheric depositions of biogenic elements and their ecological effects on marine ecosystem of Jiaozhou Bay: A review[J]. Chinese Journal of Applied Ecology, 2017, 28(1): 353-366 (in Chinese).
-



 下载:
下载: