-

图 1 MOF材料FESEM分析结果(编号同表1)
Figure 1. The FESEM result of MOF material(the number is same to Table 1)
-
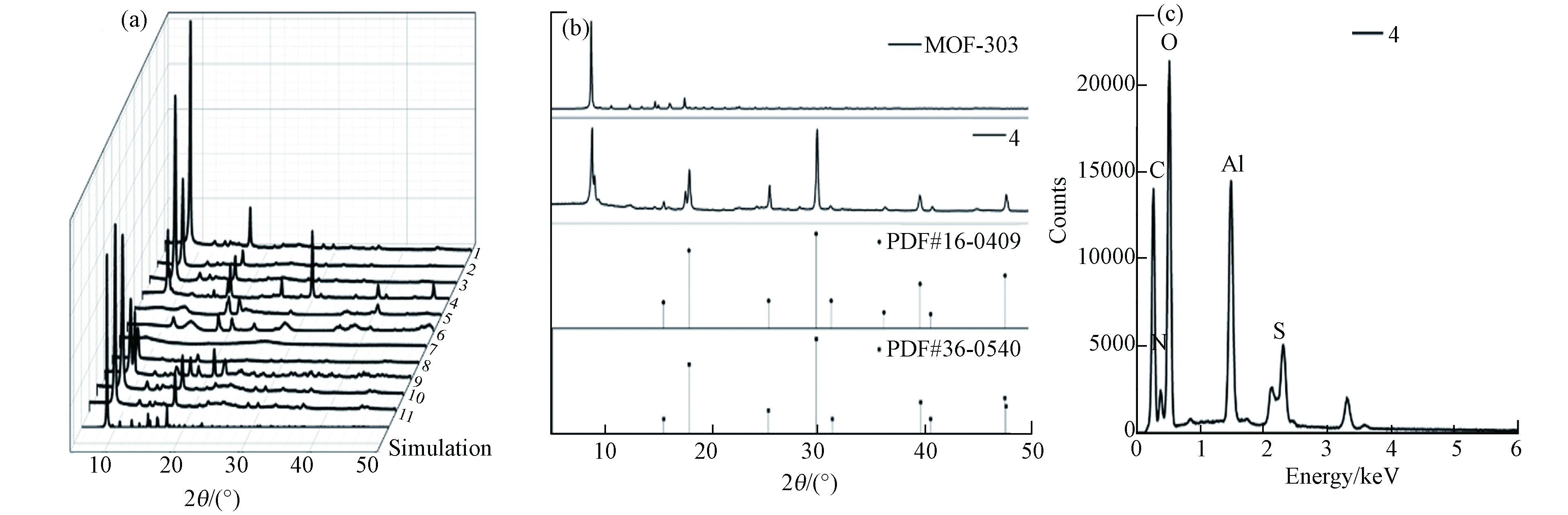
图 2 (a) MOF材料XRD曲线;(b)4号样XRD拟合结果;(c) 4号样EDX结果
Figure 2. (a) XRD curve of samples; (b) Curve fitting of sample No.4 MOF; (c) The EDX result of No.4 MOF
-
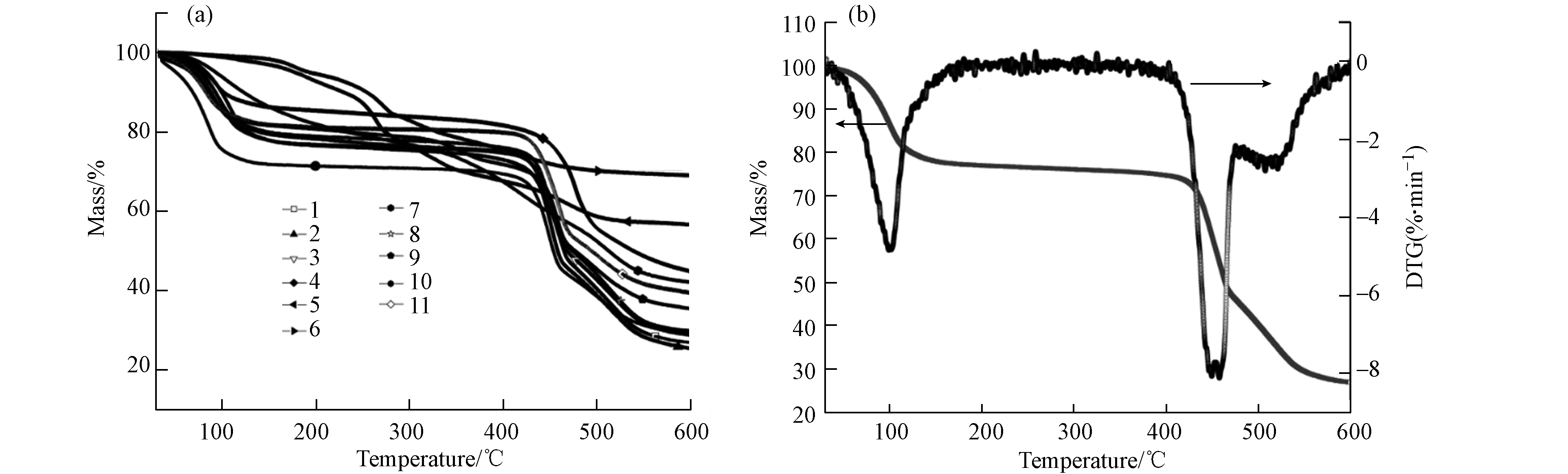
图 3 (a) MOF材料热重曲线;(b)2号材料TG-DTG曲线
Figure 3. (a) The TG curve of MOF materials; (b) The TG-DTG curve of No.2 MOF
-
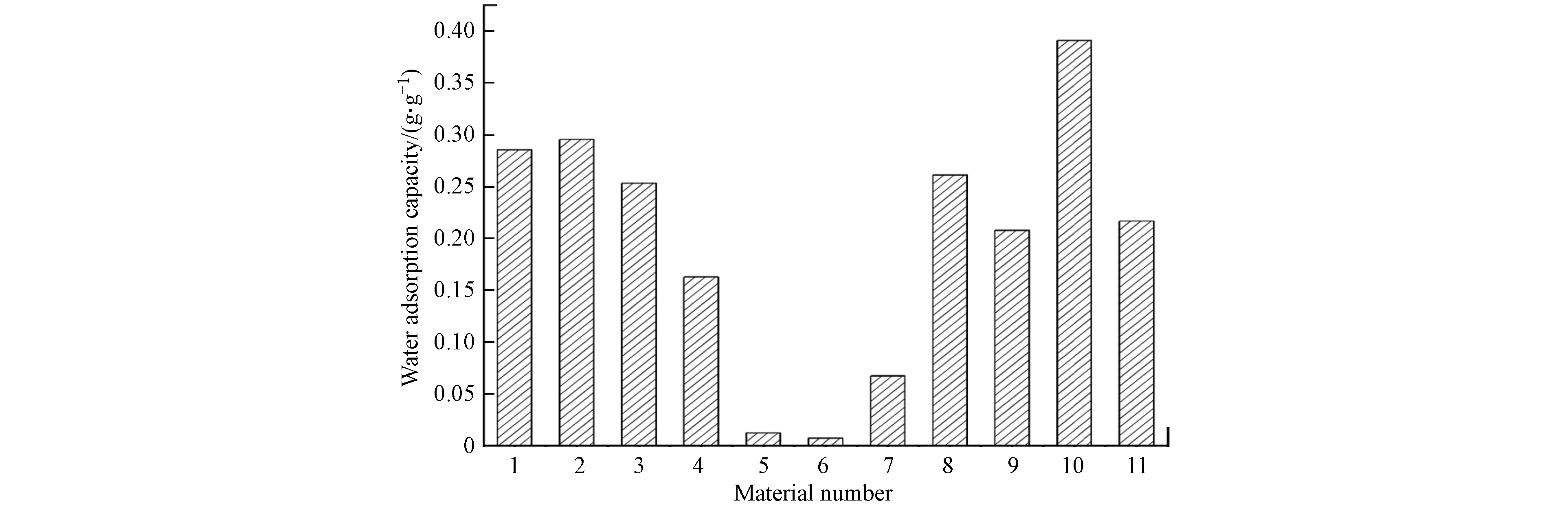
图 4 MOF材料的水吸附容量
Figure 4. Water adsorption capacity of MOF materials
-
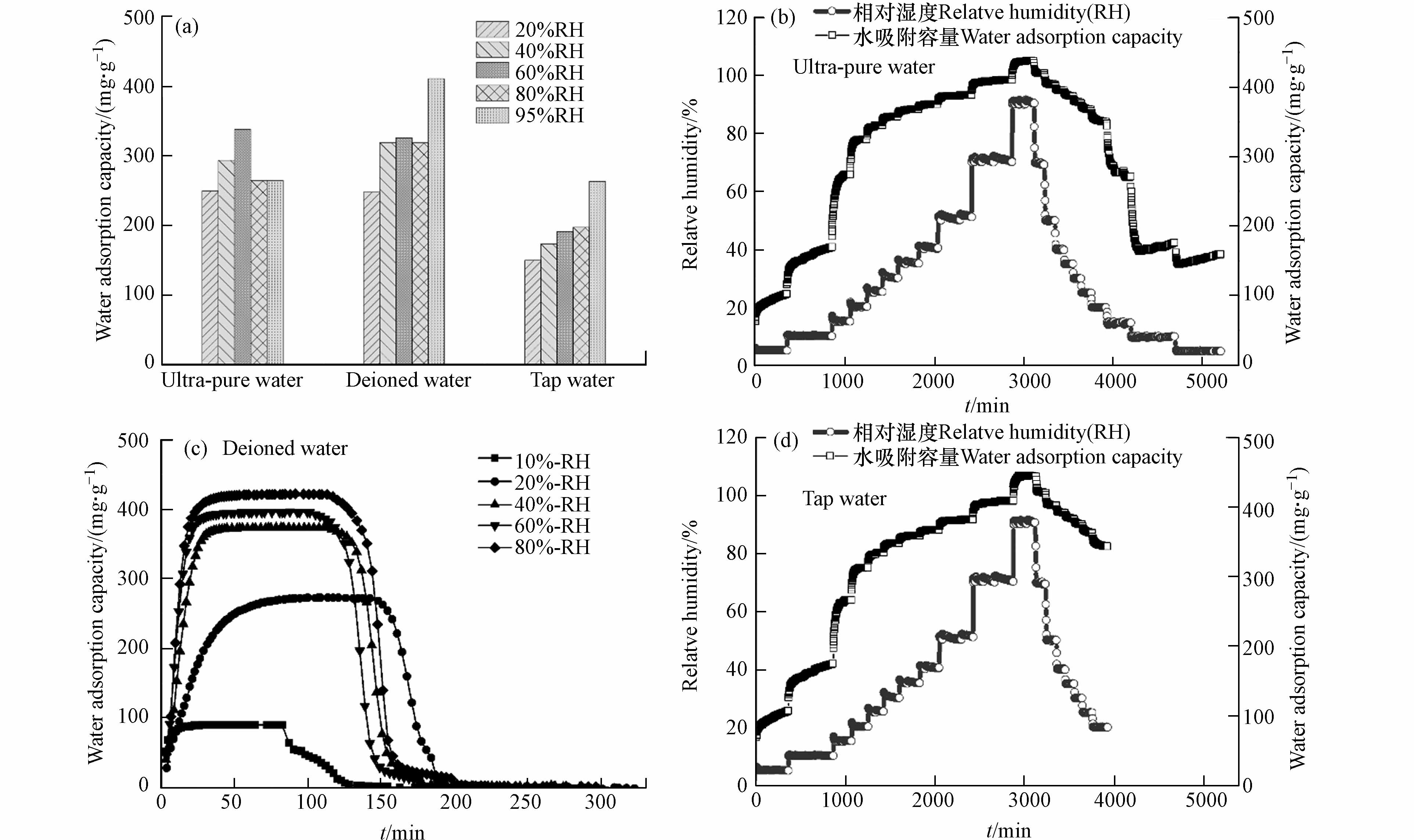
图 5 不同水溶剂下MOF材料的水吸附性能(a)不同相对湿度下的水吸附容量;(b—d)不同水溶剂下的吸附动力学曲线
Figure 5. Water adsorption performance of MOF materials under the influence of water solvent (a) water adsorption capacity under different relative humidity; (b—d) Adsorption kinetic curves under different aqueous solvents
-
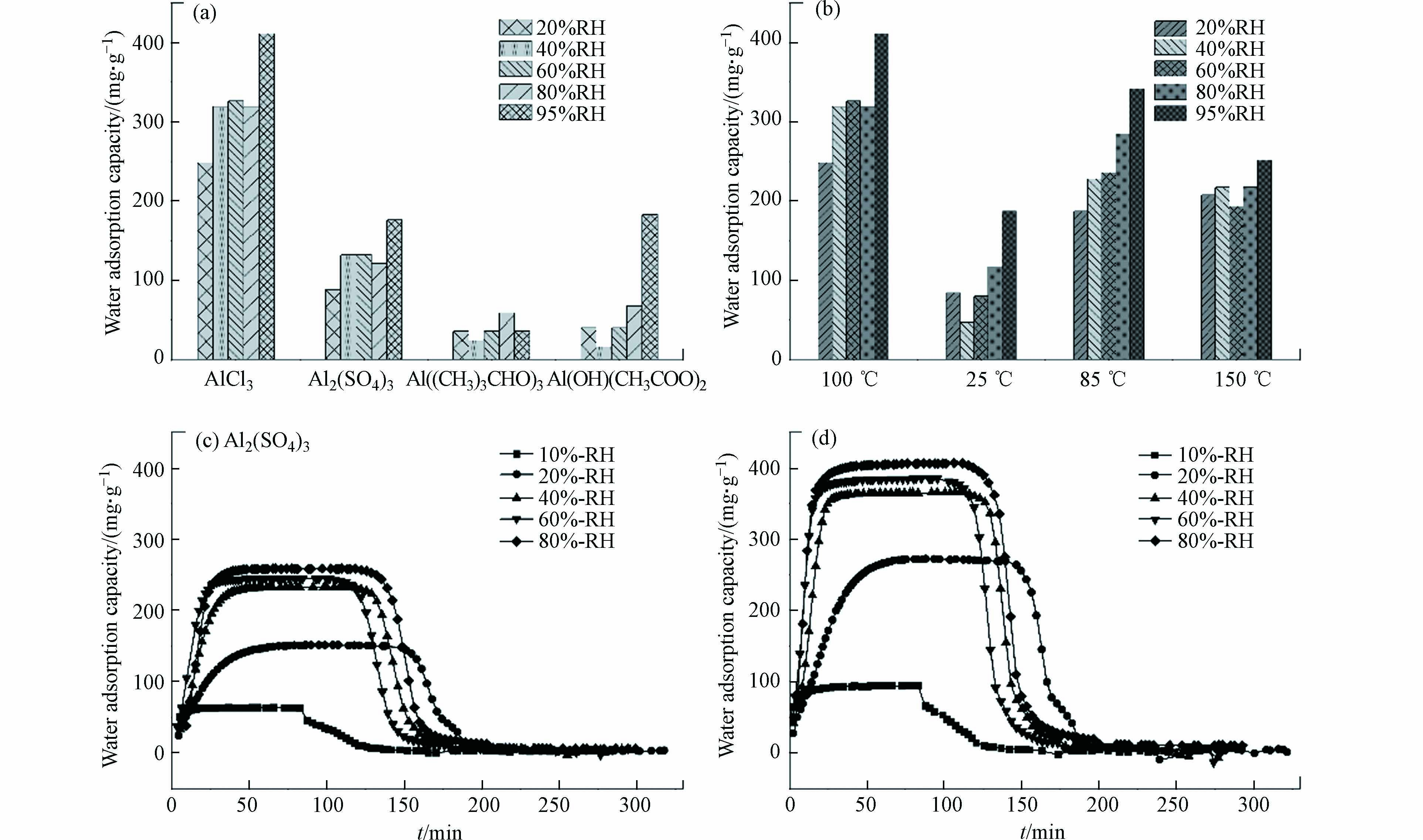
图 6 (a)不同铝源条件下制得材料的水吸附容量(样品编号:2,4—6);(b)不同温度条件下制得材料的水吸附容量(样品编号:2,7—9);(c)硫酸铝为铝源时材料吸附动力学曲线(样品编号:4);(d)85℃反应温度时材料吸附动力学曲线(样品编号:8)
Figure 6. (a)water adsorption capacity based on the influence of aluminum source (sample number:2,4—6);(b)water adsorption capacity based on the influence of temperature (sample number:2,7—9) (c)adsorption kinetic curves under aluminum sulfate as aluminum source(sample number:4) ;(d)adsorption kinetic curves under temperature of 85℃(sample number:8)
-
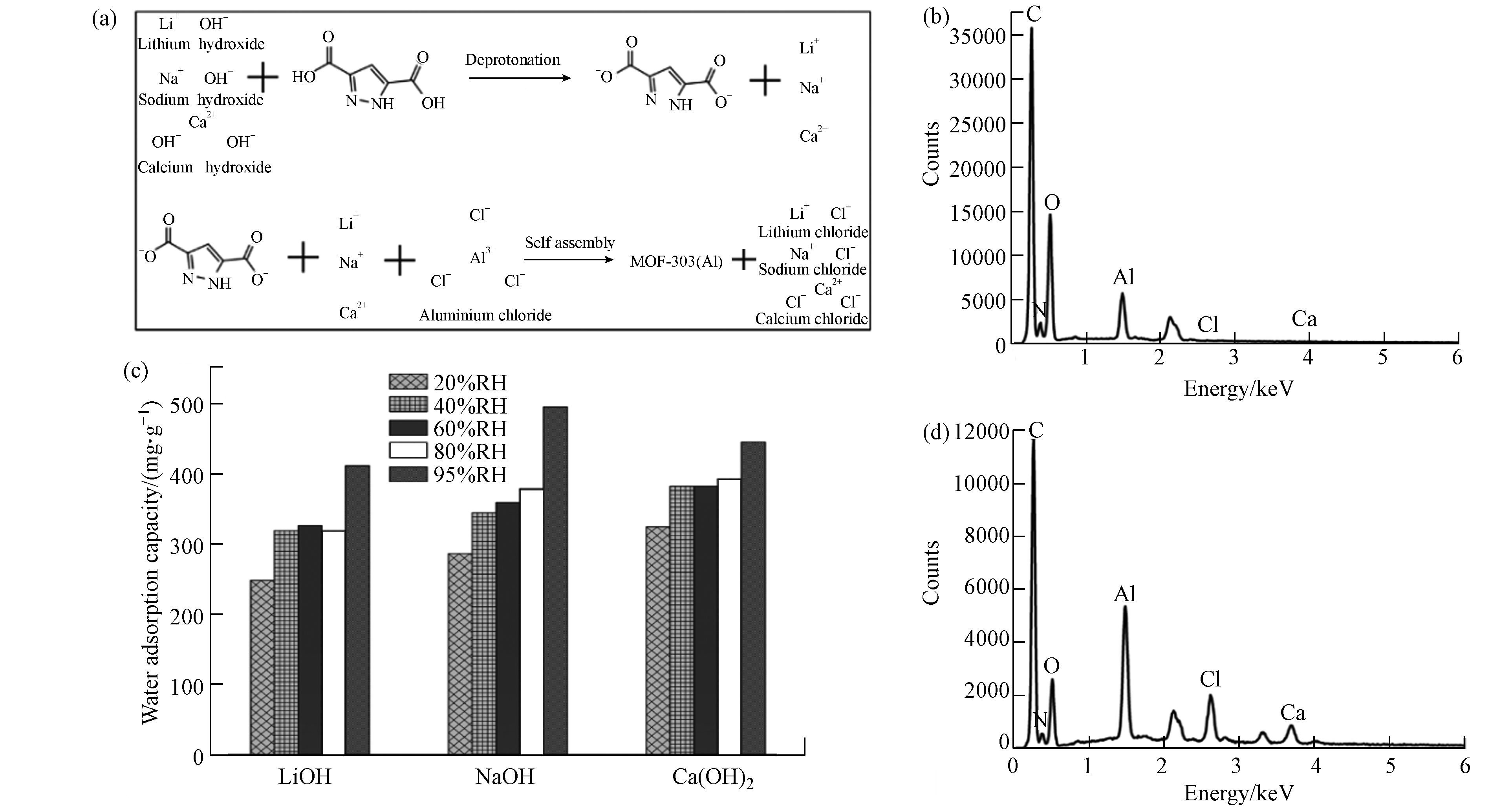
图 7 (a) MOF-303(Al)制备过程; (b) MOF材料元素分布图(氢氧化钙为分散剂,样品编号:11);(c) 分散剂影响下材料水吸附容量(样品编号:2,10-11); (d) MOF材料元素分布图(氢氧化钙为分散剂时,省略洗涤分离步骤)
Figure 7. (a) The synthesis process of MOF-303(Al); (b) Material element distribution map (calcium hydroxide as dispersant,sample number :11); (c) Water adsorption capacity based on the influence of dispersant (sample number :2,10-11);(d) Material element distribution map (calcium hydroxide as dispersant without the wash and separation step)
-
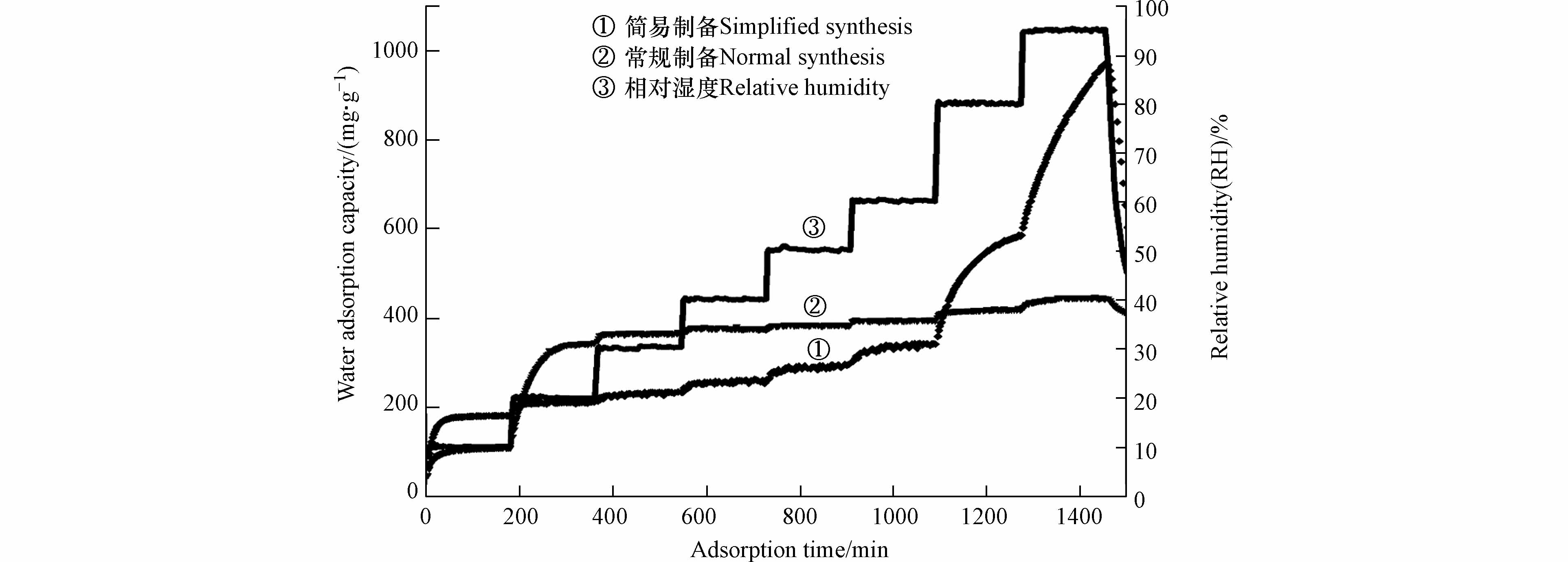
图 8 材料吸附动力学曲线(氢氧化钙为分散剂)
Figure 8. Adsorption kinetic curves of MOF (Ca(OH)2 as dispersant)
Figure
8 ,Table
4 个