-
随着药品和医疗行业的快速发展,人类和动物对抗生素的消耗量逐渐增加。抗生素可以有效治愈多种疾病,避免病情恶化[1]。常用的抗生素按照其化学结构及性质分类,包括磺胺类(sulfonamides,SAs)、四环素类(tetracyclines,TCs)、氟喹诺酮类(quinolones,QNs)、大环内酯类(macrolides,MCs)、β-内酰胺类(β-lactams,β-Ls)和氨基糖苷类(aminoglycosides,AGs)等[2]。其中,磺胺类抗生素的使用占比较高[3],磺胺嘧啶(sulfadiazine,SDZ)是一种典型的磺胺类抗生素,其制备成本较低且具有广泛的应用有效性,抗菌性强且易吸收,在人用药及兽药中被广泛使用。甲氧苄啶(trimethoprim,TMP)作为一种磺胺增效剂,常与磺胺类抗生素联用以获得更强的抗菌活性。环丙沙星(ciprofloxacin,CIP)和恩诺沙星(enrofloxacin,EFA)作为氟奎诺酮类抗菌药物,也被广泛使用。各类抗生素的广泛使用及其对生物降解的高抗性导致含有抗生素的医疗废水、生活废水无法得到有效地去除,进而在环境中不断积累。有研究表明,在陆生、水生环境,甚至直接饮用水中都已频繁检测到抗生素残留[4-5]。因此,迫切需要开发高效、环保的水处理技术,对现有的和日常输出的含抗生素废水进行有效处理。
高级氧化工艺(advanced oxidation processes,AOPs)可以产生多种具有较高氧化电位的活性氧物质,被认为是目前处理不可生物降解和难生物降解有机化合物的最有效技术[6]。在AOPs中,高活性物质既可以通过能量注入直接解离水分子产生,也可以通过不同的方式激活氧化剂生成。由于水分子的直接解离需要较高的能量消耗,而氧化剂活化方式的AOPs过程具有操作简单、能耗低的优点,从而受到广泛关注。氧化剂活化方式中常用的氧化剂有过氧化氢(H2O2)[7]、臭氧(O3)[8]、过硫酸盐(persulfate,PS)[9]、高碘酸盐(periodate,PI)[10-11]。与H2O2、O3和PS相比,PI具有更好的热稳定性,并且易于运输和储存,因此,近年来通过活化PI处理有机废水的研究备受关注[12-14]。
PI活化过程可生成多种高活性物质,包括高碘酸根自由基(IO3·、IO4·)、羟基自由基(·OH)、超氧根阴离子自由基(·O2−)、O(3P)原子、单线态氧(1O2)、电子(e−)和空穴(h+)等,其在有机物降解过程中均有一定的作用。目前已经开发并应用的PI活化方法或试剂包括冷冻[15]、超声[16]、紫外线(UV)照射[17-19]、羟胺[20]、碱[21]、金属离子[22]、非均相金属催化剂[23-26]等。TiO2作用一种广泛使用的催化剂,因其化学稳定性强、毒性小、成本效益高而被广泛研究、应用。已有研究证实,紫外光协同TiO2活化PI可以有效提高多种水体中难降解有机物的处理效果[27]。而太阳光作为可再生能源,也被用于活化PI降解水体有机污染物[28]。ZHANG等[27]使用紫外光活化PI和TiO2,用于处理水体中的有机污染物,150 min时的降解率可达88%;GUO等[29]在模拟太阳光照射下活化PI,用于降解烷基咪唑基离子液体,结果在120 min内对1-己基-2,3-二甲基咪唑溴化铵的去除率达到了99%;HUANG等[30]采用氙灯模拟太阳光活化PI,对甲氧苄啶进行降解,结果显示,初始质量浓度为20 mg·L−1的甲氧苄啶在4 mol·L−1PI的添加条件下,50 min后的降解率可达92%。
基于模拟太阳光对PI的活化作用和TiO2的促进作用,本研究建立模拟太阳光活化PI水处理体系,并在该体系中引入TiO2,系统地研究了该联合体系中典型抗生素的降解效果及过程。采用氙灯为模拟太阳(simulated solar, SS)光源,建立模拟太阳光活化PI(PI/SS)系统,首先以SDZ为目标物,考察了TiO2添加前后体系中SDZ降解效果的变化以及相应的降解过程,并通过H2O2质量浓度测定和猝灭剂添加影响实验,初步探究模拟太阳光下TiO2活化PI降解SDZ作用机理。最后,通过考察TMP、CIP和EFA等典型抗生素溶液在PI/TiO2/SS体系中的降解效果验证了该协同过程的有效性。
-
甲醇(CH4O,≥99.9%)、甲氧苄啶(TMP,C14H18N4O3,98%)、环丙沙星(CIP,C17H18FN3O3,98%)、恩诺沙星(EFA,C19H22FN3O3,98%)、乙腈(C2H3N,≥99.9%)和高碘酸钠(NaIO4,99.5%)购自上海麦克林生化科技有限公司。氢氧化钠(NaOH,≥96%)、盐酸(HCl, 36.0%~38.0%)、甲酸(CH2O2,≥98%)、异丙醇(C3H8O,≥99.7%)、对苯醌(C6H4O2,≥98%)、叔丁醇(C4H10O,≥99.0%)、苯酚(C6H6O,≥98%)均来自国药化学试剂有限公司。磺胺嘧啶(SDZ,C10H10N4O2S,98%)、糠醇(C5H6O2,≥98%)、DMPO(C6H11NO,≥97%)、TEMP(C9H19N,≥98%)和β-胡萝卜素(C40H56,≥96%)购自上海阿拉丁生化科技有限公司。制备溶液所用的水均为去离子水。
-
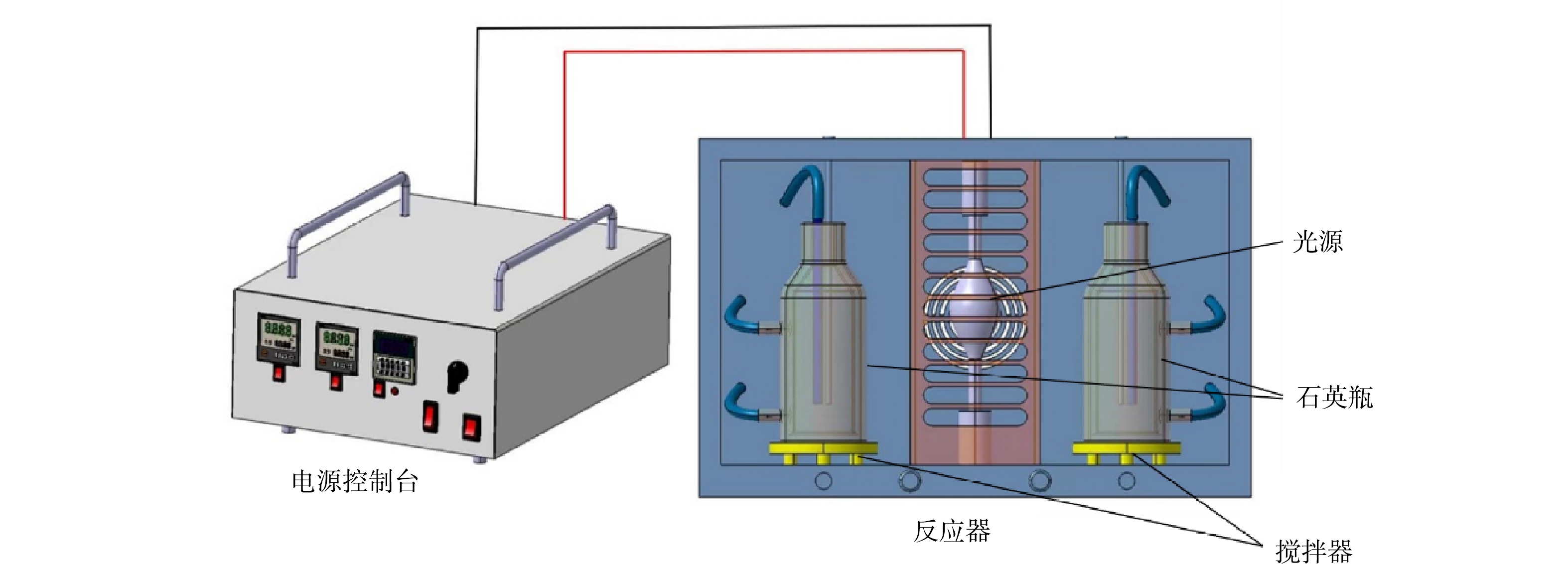
抗生素降解实验在GHX-3光化学系统(购自扬州大学城科教仪器有限公司)中进行,装置如图1所示。装置主体由电源控制台和反应器2部分组成。电源控制台用于开关光源、控制冷却循环水以及设定反应温度。反应器内对称放置有2台磁力搅拌器,搅拌器之间为光源。实验过程中,将盛有目标溶液的双壁石英瓶(容积为500 mL)放置于搅拌器上,反应过程中不断有冷却水流过石英瓶的夹层,保证瓶内反应温度恒定为25 ℃,采用250 W的氙灯作为模拟太阳光源。
-
实验中目标物的初始质量浓度为20 mg·L−1(溶液初始pH约为6.9),处理量为300 mL,反应温度控制在25 ℃。每隔10 min取出样品,通过0.22 μm膜过滤,用于后续分析。通过0.1 mol·L−1 NaOH溶液和0.1 mol·L−1 HCl溶液调节溶液的初始pH。每种条件下的所有实验均重复3次。
-
使用配置有紫外检测器的高效液相色谱仪(HPLC)测定溶液中抗生素的质量浓度,配套的色谱柱为Symmetry C18(4.6 mm×250 mm,5 μm)柱,测定SDZ、TMP、CIP和EFA的检测波长分别为269、271、277和276 nm,流动相流速和进样量分别为1.0 mL·min−1和20 μL。采用液相色谱-质谱法(LC-MS)对SDZ的降解产物进行鉴定。鉴定前的固相萃取(SPE)流程如下:样品溶液注入活化后的色谱柱(Waters C18),而后进行洗脱,收集的洗脱液在氮气气氛下干燥后溶解于初始流动相中进行LC-MS测试。测试条件:流动相为0.1%甲酸(A相)和甲醇(B相)(70:30),进样量5 μL,流速0.3 mL·min−1。质谱仪采用电喷雾电离(ESI)源,离子源参数设置为:护套气温度350 ℃,护套气流量12 L min−1,检测电压4 000 V。
根据测定的有机物浓度计算其降解率,并进行相应的准一级动力学分析。采用硫酸氧钛法测定H2O2的质量浓度[31];使用北京连华科技公司的COD检测仪测定降解过程中溶液的COD值,并计算去除率;采用紫外可见分光光度计(UV-2 600,岛津,日本)对降解过程中的目标物溶液进行紫外-可见全波段扫描分析。
-
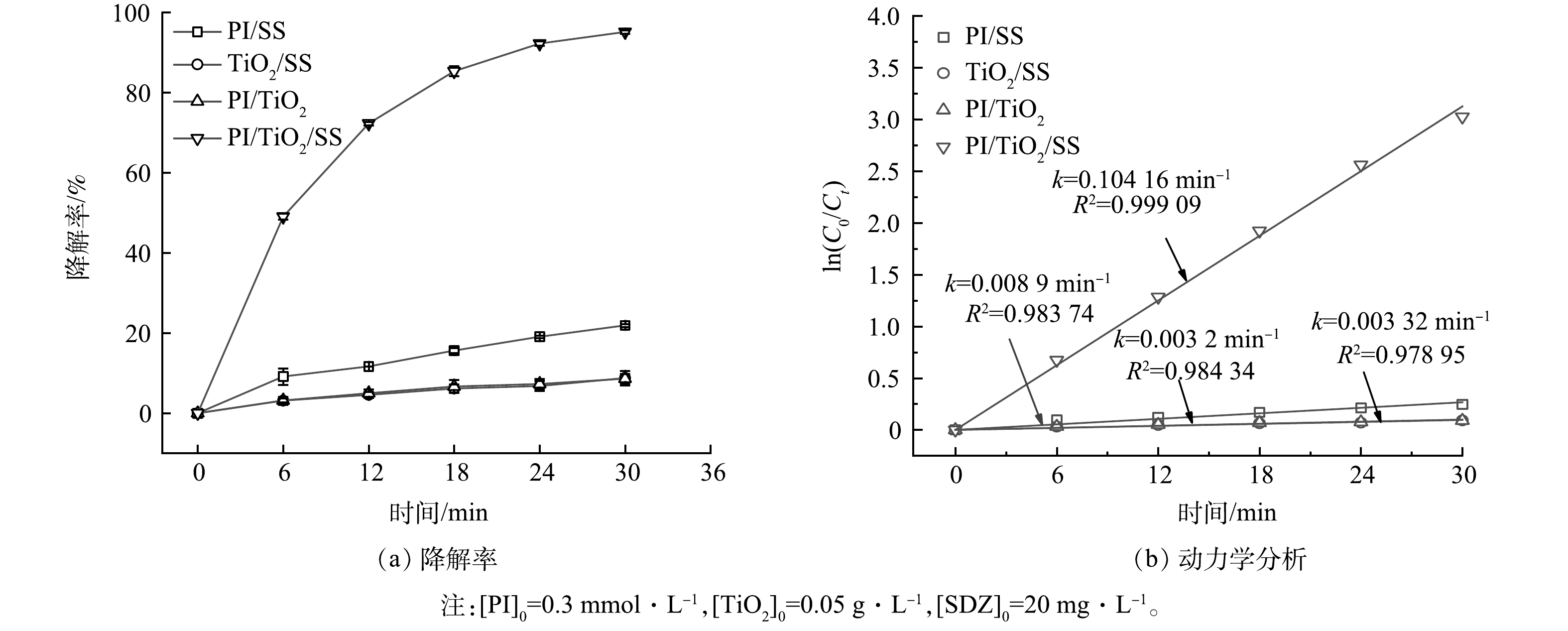
研究首先比较PI/SS、TiO2/SS、PI/TiO2和PI/TiO2/SS四个体系中SDZ降解效果的变化,以证实SS、TiO2与PI三者对SDZ的协同降解作用,结果如图2所示。由图2(a)可以看出,在SS/TiO2和PI/TiO2体系中,反应30 min后SDZ的降解率分别为8.76%和8.60%,说明SS和PI分别结合TiO2时对SDZ的降解效果较差。在PI/SS体系中,处理30 min的SDZ的降解率为21.90%,说明PI/SS体系中SDZ的降解效果有限且模拟太阳光对PI有一定的活化作用,即氙灯可以在一定程度上激活PI [29],是使PI变为活化状态(PI*),并通过PI*与溶液分子的反应生成许多具有高电位的活性物质,从而氧化SDZ。活化过程中各种反应物质的形成如下(式(1)~式(11))[27]。在PI/TiO2/SS体系中,30 min内对SDZ的降解率为95.13%,说明PI、TiO2和SS三者对SDZ降解具有高效的协同作用。图2(b)为动力学分析结果。在PI/TiO2/SS体系中的动力学分析常数分别是PI/TiO2、PI/SS和TiO2/SS体系的31.37、11.70和32.55倍。该研究结果证实了PI、TiO2和SS三者的协同作用。其原因可能为:在水溶液中,PI吸收光后,电子从基态HOMO被激发到激发态LUMO,进而生成多种活性物质,作用于有机物;TiO2吸收光后,部分电子从价带 (VB) 激发到导带 (CB) ,产生电子和空穴对,该电子可以与水中的溶解氧发生反应,生成·O2−,进而生成·OH[32],氧化目标物;而PI所具的强正电荷还可以有效捕获TiO2上的价带电子,从而抑制了电子-空穴对的复合,继而形成协同效应,强化有机物降解效果。该协同过程的作用过程如式(12)~式(17)[33]。
考虑到PI/TiO2/SS体系中影响有机物降解的3个关键参数:TiO2添加量、PI添加浓度和溶液pH。通过正交实验优化PI/TiO2/SS体系中这3个参数,其中TiO2添加质量浓度设为A,PI添加量设为B,pH设为C,处理SDZ溶液初始质量浓度为20 mg·L−1,水量为300 mL。正交实验设计参数列于表1,相应不同反应体系中30 min后SDZ的降解率列于表2。表中的K1、K2和K3为不同的影响因子在不同水平下磺胺嘧啶的降解效率之和,例如:K1表示1水平下磺胺嘧啶的降解率之和;k1、k2和k3为不同的影响因子在不同的水平下磺胺嘧啶的降解效率和的平均值,例如:
$ {k}_{1}=\dfrac{{K}_{1}}{n} $ ,其中n=3;R为同一个影响因子下不同水平的极差值,即R=kmax-kmin,其中kmax和kmin分别为同一个影响因子下不同水平的最大值和最小值。由表2结果可以看出,SDZ的降解率随PI浓度的增加而大幅增加,TiO2质量浓度和pH变大的情况下,SDZ的降解率的变化则很小。有关PI浓度的影响结果,是因为随着PI浓度的增加,PI分子的入射光捕获机会增加,然后导致SDZ降解的活性物质增加。当加入的TiO2越多,虽然提供的反应表面变大,但PI的含量是一定的,故而产生的电子(e−)和空穴(h+)的总量是一定的,不会因为TiO2的增加而大幅度增加。对于溶液pH的影响规律,在pH>8时,四面体高碘酸阴离子(IO4−)占主导地位,而在pH<8时,二聚体H2I2O104−最为常见(式(18)~式(21))。研究表明,H2I2O104−与IO4−具有相似的氧化还原电位[17],所以溶液pH对SDZ的降解影响不大。
注∶K1、K2和K3为不同的影响因子在不同水平下磺胺嘧啶的降解效率之和,k1、k2和k3为不同的影响因子在不同的水平下SDZ的降解效率和的平均值,R为极差值。
综合表2中的SDZ降解结果,可以得出PI/TiO2/SS体系降解SDZ的较优操作条件为:PI浓度0.3 mmol·L−1、TiO2质量浓度为0.15 g·L−1、pH为9。再通过极差R的分析:PI浓度(34.99 mmol·L−1)>TiO2质量浓度(1.90 g·L−1)>pH(1.47),可得出以上3种影响因素中PI浓度对SDZ降解效率影响最大,TiO2质量浓度和pH的影响较小,从考虑实验操作的简便性和经济性,本实验考虑选择较低的TiO2质量浓度,而对溶液的初始pH不做调节,即保持SDZ溶液的初始pH条件。
-
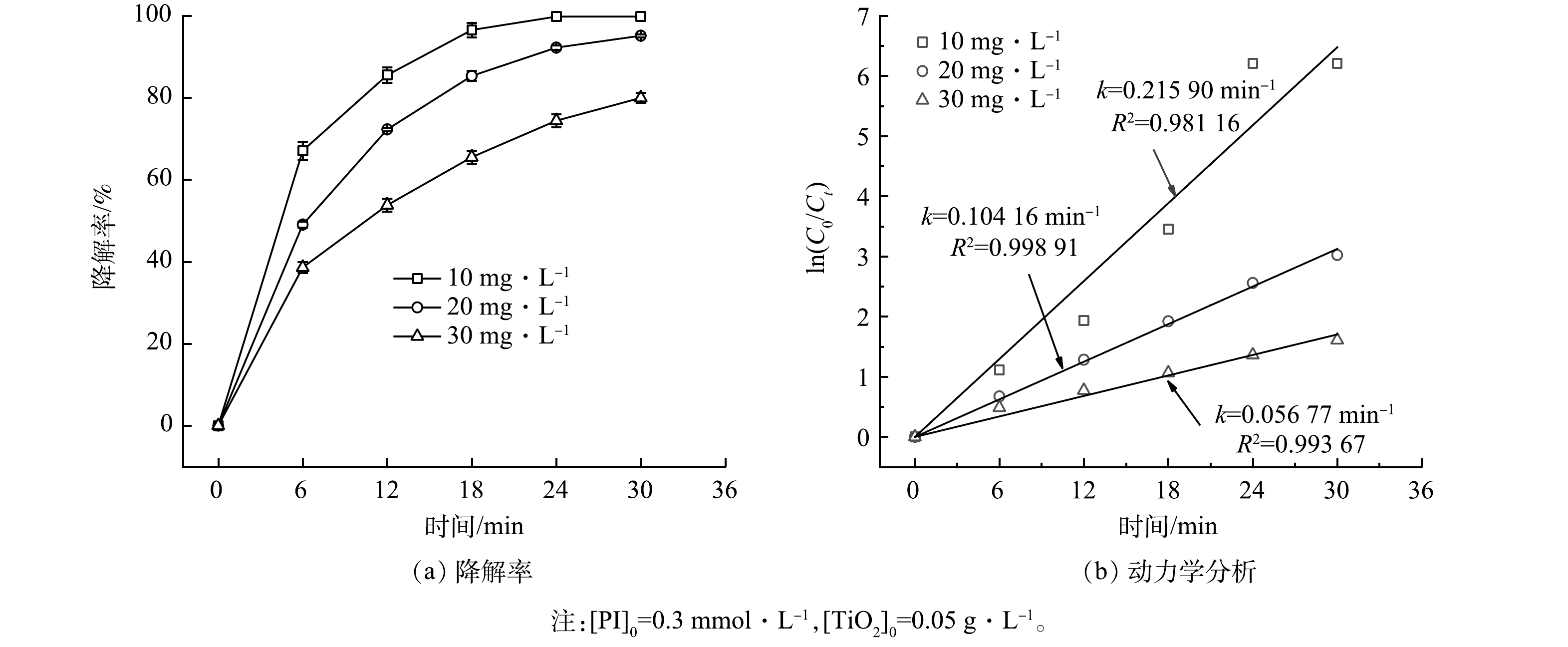
1)磺胺嘧啶初始质量浓度的影响。考察了不同初始质量浓度(10、20、30 mg·L−1)的SDZ对PI/TiO2/SS体系中SDZ降解的影响,结果如图3所示。由图3可见,在30 min时,10、20、30 mg·L−1 SDZ的降解率分别为99.8%、95.13%和79.99%;k值也从10 mg·L−1的0.215 90 min−1降至30 mg·L−1的0.056 77 min−1。该结果表明,SDZ初始质量浓度越低,降解速度越快。其原因可能为以下4点:1)当PI和TiO2为固定量下,固定氙灯功率,产生的活性物质数量基本一致,而随着SDZ初始质量浓度的增加,对于活性物质的需求量增加,产生的活性物质相对不足[33],即每摩尔SDZ对应的活性物质减少,导致降解率降低[17];2)SDZ初始质量浓度越大,降解开始时产生的副产物也越多,其会与SDZ本身形成竞争反应,影响SDZ的降解;3)SDZ质量浓度越高,对光的吸收也越多,导致PI和TiO2可利用的光减少,从而削弱PI的活化效果和SDZ的降解;4)当SDZ初始质量浓度增加时,目标分子在TiO2表面的占比会增加,导致SDZ分子覆盖了TiO2的活性位点[27],从而降低了TiO2表面的活性物质的生成量,导致SDZ降解率降低。
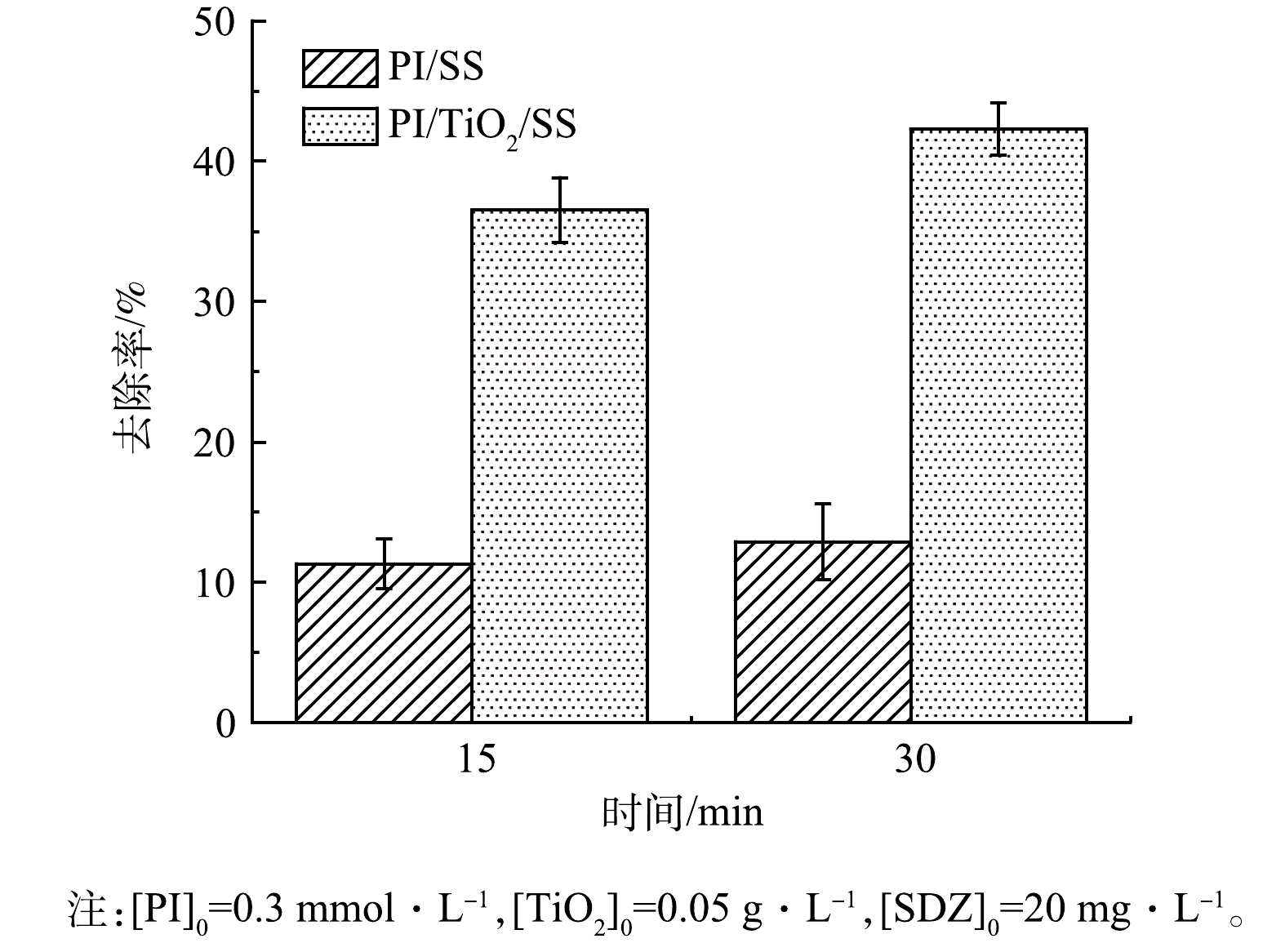
2)溶液COD变化。考察了光照和PI浓度条件一致时,添加0.05 g·L−1的TiO2后,SDZ溶液COD的去除效果变化。由图4可知, PI/SS和PI/TiO2/SS 2种反应体系中COD的去除率均随反应时间的延长逐渐升高。在PI/SS体系中,30 min后溶液中COD的去除率为12.9%,在PI/TiO2/SS体系中,30 min后溶液中COD的去除率为42.3%,提高了29.4%,说明TiO2的加入可以有效提高溶液中耗氧有机污染物(以COD计)的降解。
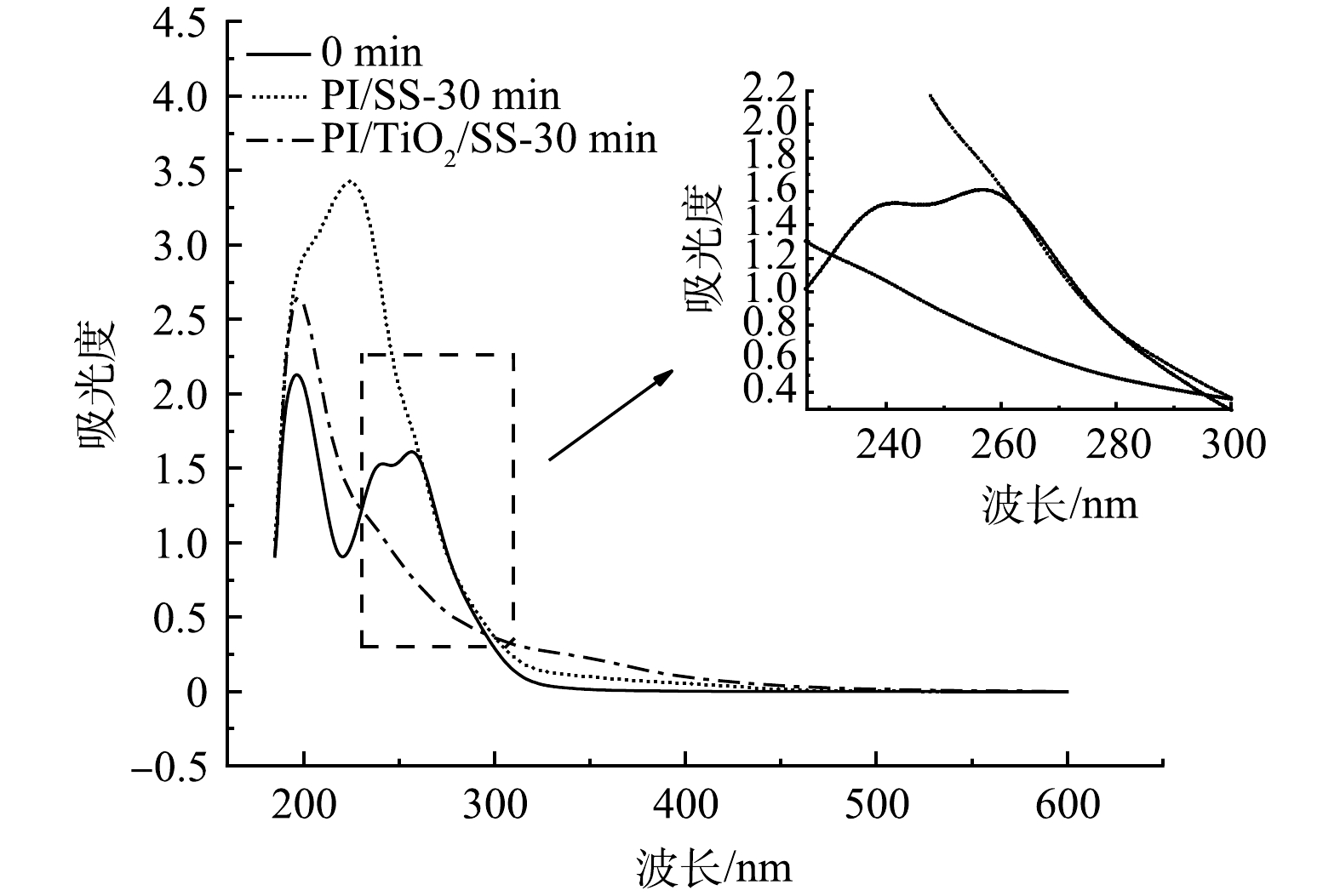
3)溶液紫外-可见全波段扫描分析。研究考察了光照和PI浓度条件一致时,添加0.05 g·L−1的TiO2前后,SDZ溶液降解30 min后紫外-可见全波段扫描图与初始溶液全波段扫描图的比较结果如图5所示。从图5可以看出,反应30 min后,经两种体系处理后溶液的紫外-可见全波段光谱变化趋势明显不同。根据SDZ的0 min的UV-Vis光谱图,在波长200~500 nm的范围内一共扫描出两个吸收峰,分别在200 nm和250 nm附近,其中,200 nm附近的吸收峰是由SDZ上的苯环结构所致,经不同体系处理后,溶液的特征峰发生变化,在215 nm处出现了一个较大的吸收峰,并且吸收强度提高,呈现红移的趋势,这是因为苯环上发生了-OH和-NH2取代。这是由于SDZ的降解,产生了大量携带苯环结构的小分子产物,这些小分子物质容易在·OH的作用下取代苯环产生-OH,导致215 nm处的吸收峰的红移。比较发现,在180~280 nm波长范围,经PI/TiO2/SS处理的SDZ溶液吸光度较PI/SS体系处理的低,说明联合体系对SDZ降解中间产物的进一步矿化的程度更高。该结果同样证实了PI、TiO2和SS三者的协同作用。
-
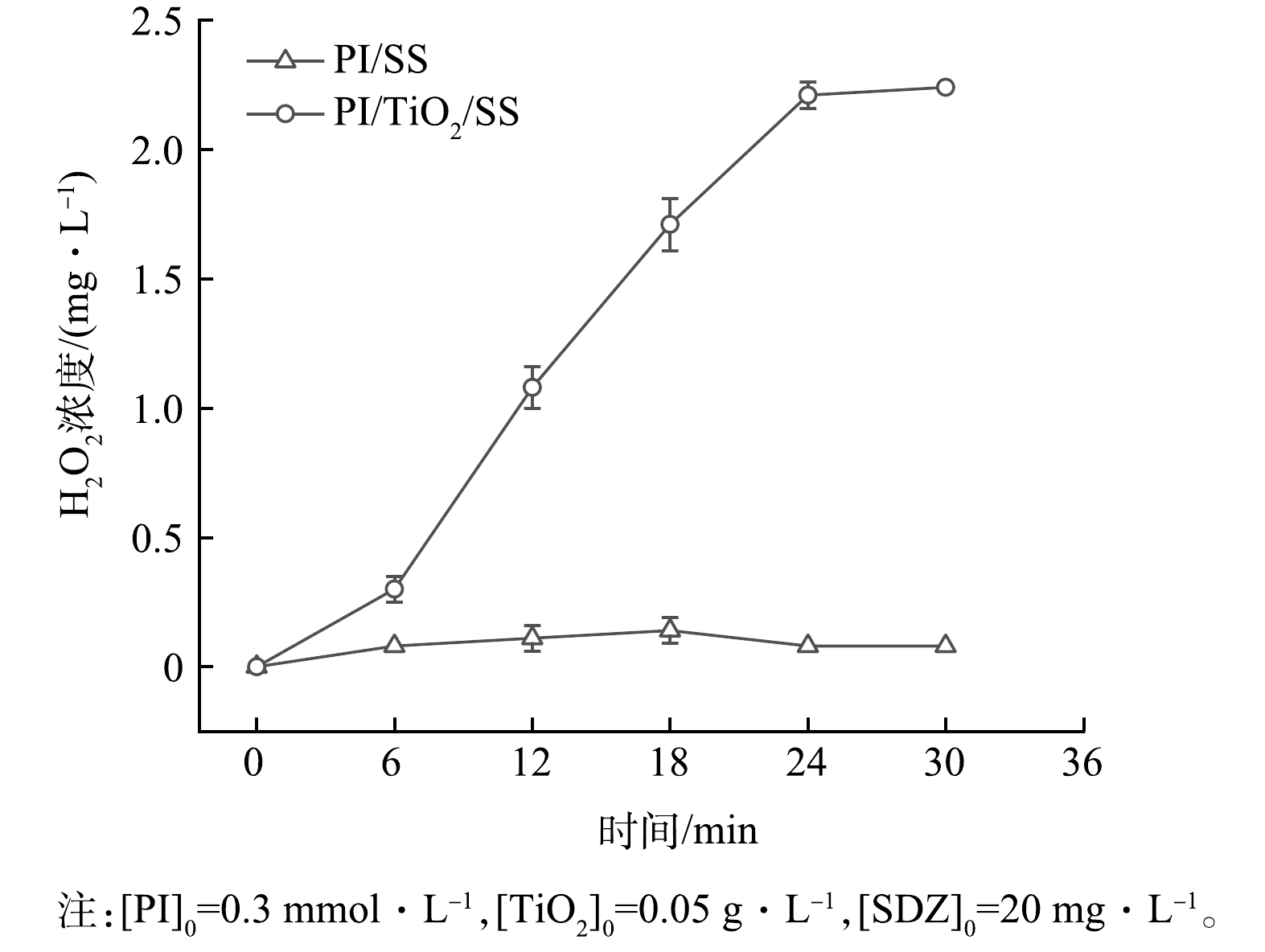
1)H2O2质量浓度分析。为了初步探究PI/TiO2/SS协同体系降解SDZ作用机理,研究考察了溶液为去离子水的条件下,PI/SS和PI/TiO2/SS 2个体系的水溶液中产生的H2O2的质量浓度变化,其他实验条件与2.3一致。由图6可见,PI/SS体系中H2O2质量浓度随反应时间的延长波动不大,PI/TiO2/SS体系中H2O2质量浓度随着反应时间的延长而增加,说明在PI/TiO2/SS体系中H2O2更容易积累。这是因为TiO2的加入使溶液中产生更多的·O2−,从而和水反应产生更多的H2O2(式((10)和式(16)),从而利于体系中有机物的降解。
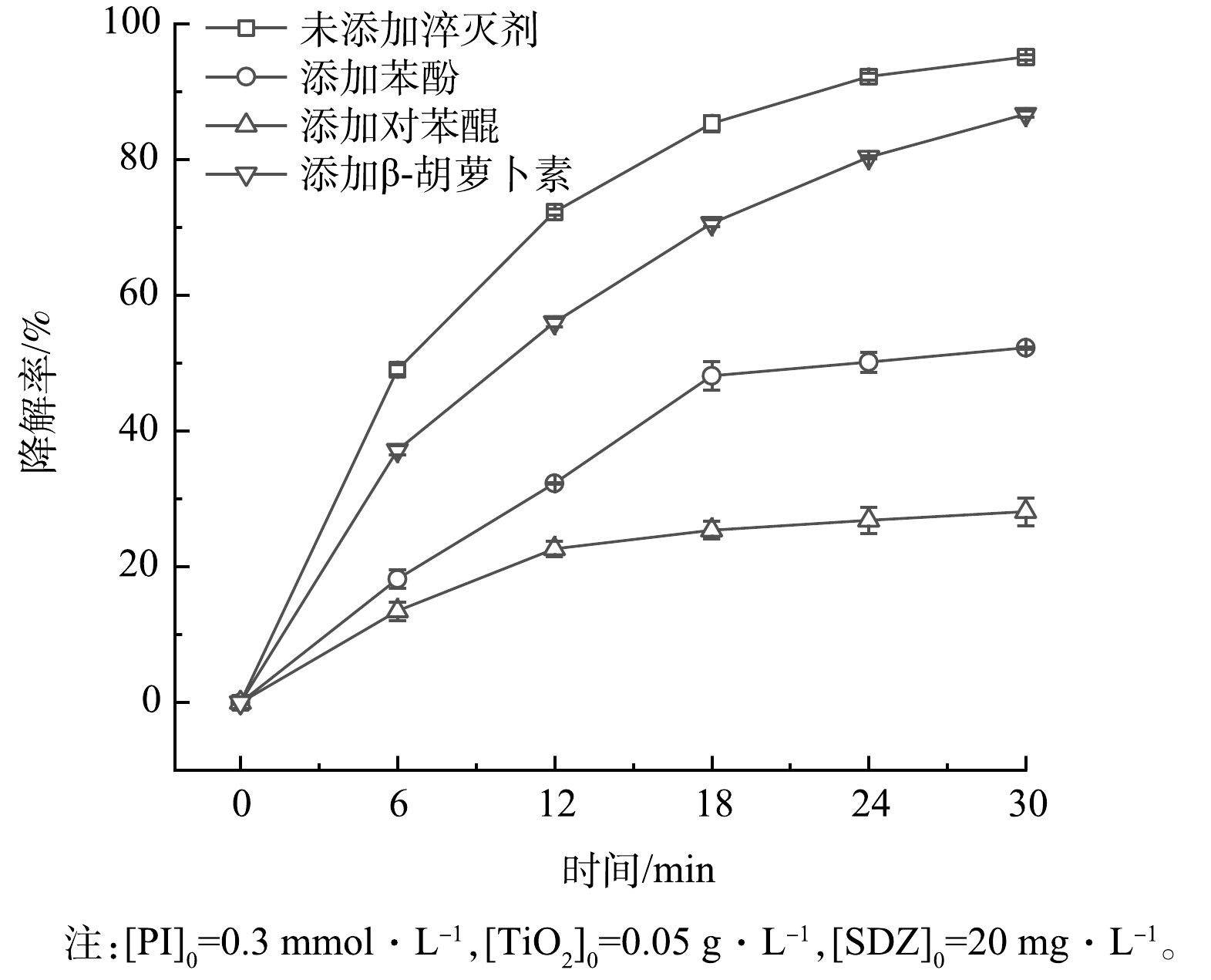
2) 猝灭剂添加实验。为了说明PI/TiO2/SS体系中产生的各种活性物质对SDZ降解的贡献,研究考察了不同猝灭剂添加对SDZ降解的影响。研究中选择的猝灭剂包括苯酚(清除IO3·、IO4·和·OH)、对苯醌(BQ,清除·O2−)、β-胡萝卜素(清除h+)[27, 34],添加量均为1 mmol·L−1。由图7可见,添加苯酚、对苯醌和β-胡萝卜素后,反应体系中SDZ去除率明显下降,反应30 min后,SDZ降解率分别由95.13%(无猝灭剂)下降到52.27%(添加苯酚)、28.12%(添加对苯醌)和86.72%(添加β-胡萝卜素),抑制顺序为:对苯醌>苯酚>β-胡萝卜素。该结果表明,在PI/TiO2/SS体系中,·O2−对SDZ的降解具有较高的影响,IO3·、IO4·和·OH对SDZ的降解也有显著的影响,而h+对SDZ降解的贡献相对较弱。这是因为在反应体系中,e−易和O2发生反应而生成·O2−(式(2)、式(13)、式(14)),使得溶液中·O2−含量相对较高从而在降解SDZ过程中起主要作用。光照PI产生的h+在溶液中和水进一步反应生成·OH(式(3))。在这一过程中,h+属于过渡性活性物质,对降解SDZ起着重要作用是后续生成的·OH。此外,IO4·已被证明对有机化合物的氧化降解作用微小[18, 20, 35],由此说明,IO3·和·OH的作用相对重要。综上,PI/TiO2/SS体系中产生的不同活性物质对有机物降解的贡献大小为:·O2−>IO3·和·OH>h+。
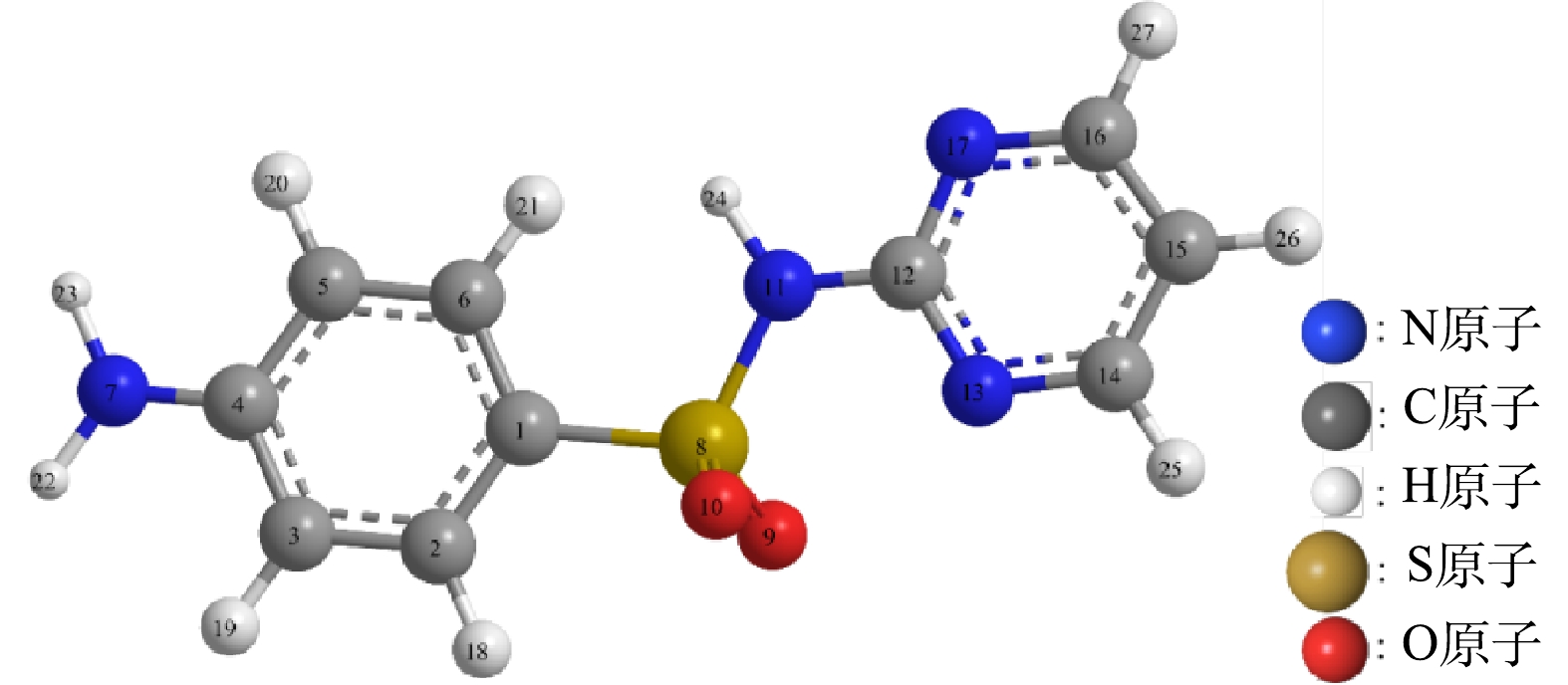
3) SDZ降解中间产物分析。根据ZHU等和YAO等[36-37]报道的SDZ的Fukui函数分析得知,SDZ受自由基攻击较为明显的是N7、N11、C1、C3、C5、N13、N17、C14和C16(图8)。可见SDZ的苯环、氨基和嘧啶杂环易受自由基攻击,但由于苯环和嘧啶杂环由于π键的离域而非常稳定,N7与C4原子之间、N11与C12原子之间的键也比较强,而C1与S8原子之间、S8与N11原子之间的键则比较脆,故而,SDZ易发生氨基氧化、S-N键断裂和脱硫反应。
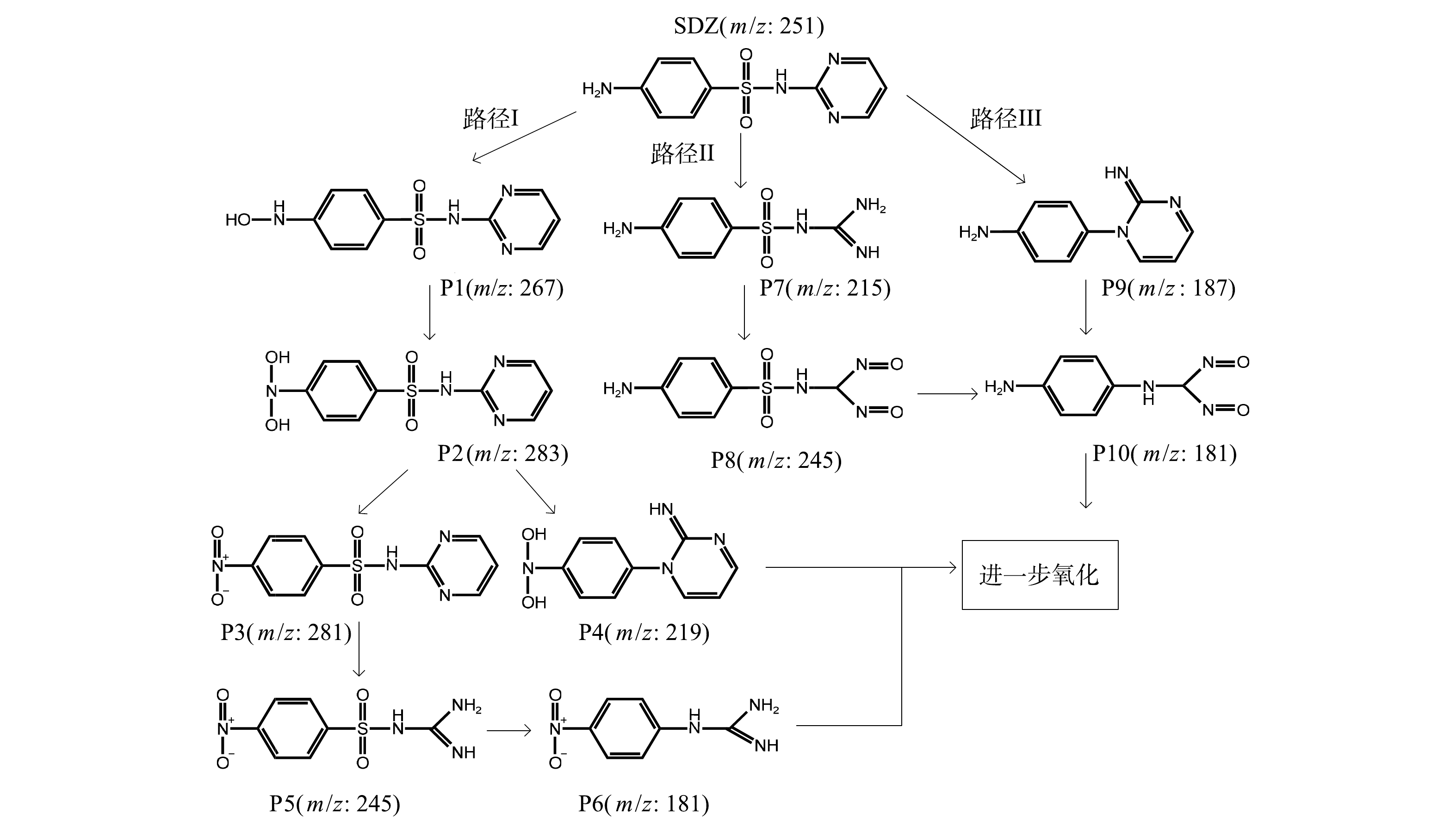
基于通过LC-MS检测、鉴定出的SDZ降解中间产物结合,结合文献,归纳出本系统中SDZ的3种可能降解路径(图9)。路径I:由于N7原子受到活性物质(·OH、IO3·、IO4·和1O2等)的作用,发生脱氢取代反应,生成产物P1和P2;随着活性物质持续作用于N-OH键,导致氨基逐渐氧化为硝基,生成产物P3;嘧啶环的N-C键在活性物质的攻击下形成产物P5;活性物质破坏N11-S8键和S8-C1键,引起脱硫反应,使磺胺上的硫原子以硫酸盐的形式释放出来,苯环和嘧啶环继而重新连接[38-39],磺酰胺在活性物质作用下形成了P6;P2上的磺酰胺受到活性物质的作用,导致S8-N11键断裂,并经过Smiles重排反应生成产物P4。路径Ⅱ:嘧啶杂环可以被·O2−攻击和破坏,生成P7;氨基进一步被等活性物质氧化生成P8。路径Ⅲ:自由基的持续攻击导致C1-S8和N11-C12键断裂以及嘧啶杂环电子分布的改变,形成P9,P9经分子重排后可形成P10。
-
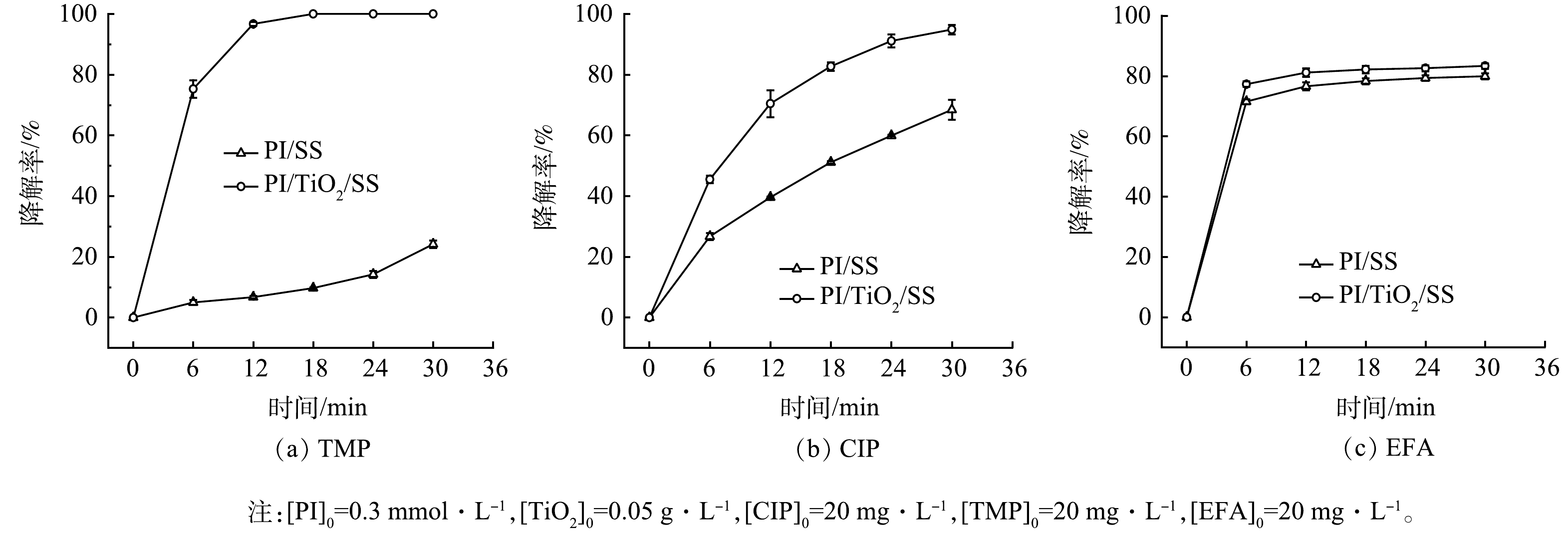
研究考察了甲氧苄啶(TMP)、环丙沙星(CIP)和恩诺沙星(EFA)在PI/TiO2/SS体系中的降解效果。这些有机物分子可以被细分为分子中具有供电子取代基(-OH,-NH2)的分子(TMP和EFA)和分子中具有吸电子取代基(卤素(-X))的分子(CIP)。所得结果如图10所示。该研究中PI用量为0.3 mmol·L−1,TiO2用量为0.05 g·L−1。结果表明,几种目标污染物在PI/TiO2/SS体系中的降解效果均比在PI/SS体系中好,并且污染物在PI/TiO2/SS体系中反应30 min内的去除率为80%~100%。其中,CIP和TMP在PI/TiO2/SS体系中表现出极高的降解率。上述结果表明,PI/TiO2/SS体系可以高效降解废水中含有不同官能团的抗生素。
-
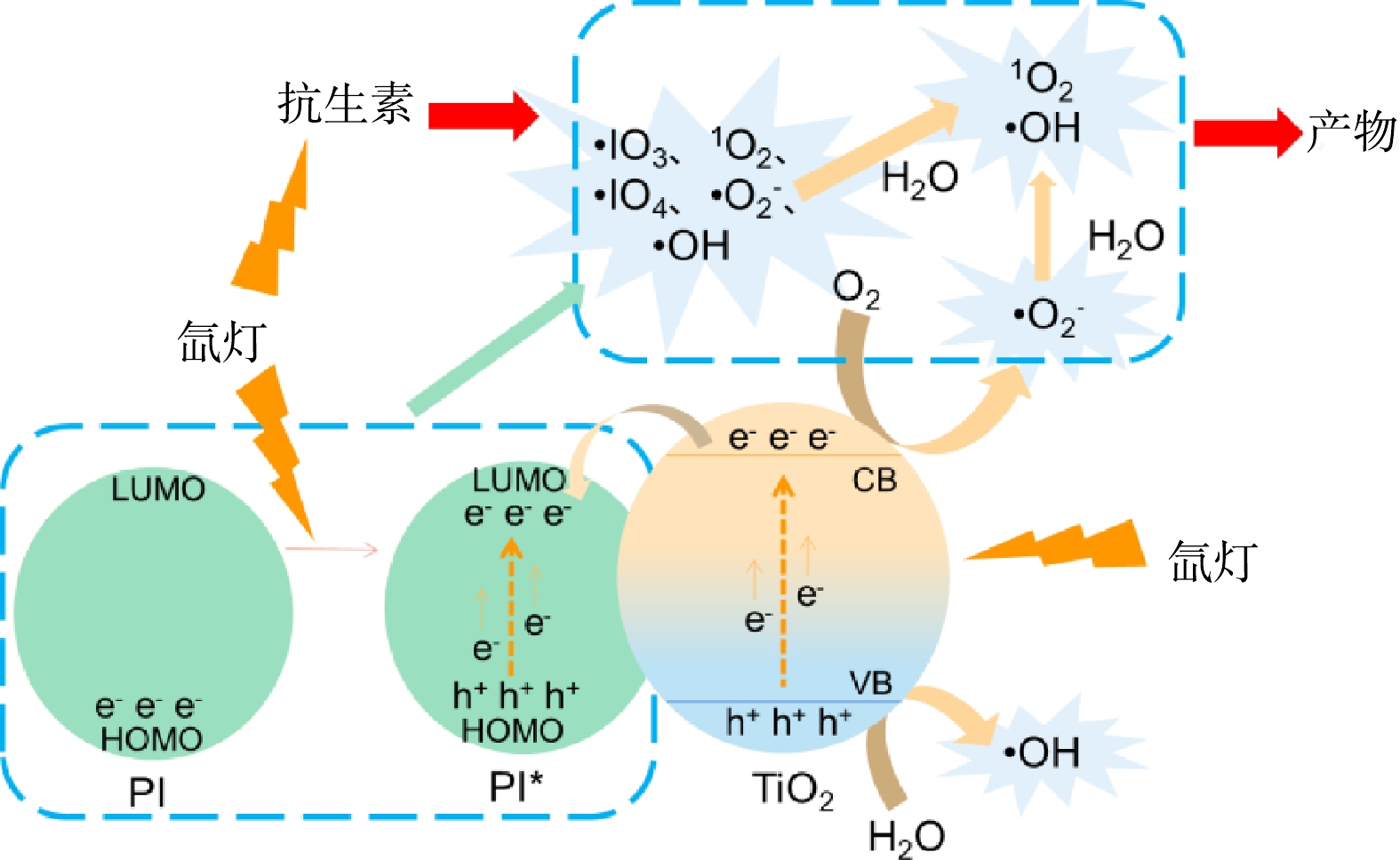
根据以上的研究,归纳模拟太阳光下TiO2活化高碘酸钠降解水体抗生素的机理如图11所示。在太阳光照射下,高碘酸钠可以吸收光子,使电子从HOMO能级激发到LUMO能级(PI转变为激发态PI*)。之后,PI*的I-O键断裂生成·O−和IO3·,而·O−与H2O反应生成·OH。在该活化过程中,还可以生成IO3−和O(3p)。HOMO中的e−可以与液相中的O2反应生成·O2−和1O2,LUMO中的h+可以与H2O反应生成·OH,并引发其他反应(式(1)~式(11))。与此同时,TiO2原子吸收光能后,其部分电子会从价带(VB)激发到导带(CB),产生电子-空穴对,该电子和空穴可以与O2及H2O反应生成各种活性物质(式(12)~式(17))。TiO2产生的电子具有较高的能量,可以从TiO2的CB转移到PI,从而抑制了其自身电子-空穴对的复合,进而提升了光生载流子的利用效率,提高反应体系中有机物的降解效果。此外,被吸附在TiO2表面的抗生素,将易于受到各种活性物质的攻击,促使其降解。
-
在本研究所建立的模拟太阳光下TiO2活化PI降解抗生素体系中,证实了PI、TiO2和SS三者对抗生素降解的协同作用。主要结论如下:
1)在反应30 min时,PI/TiO2/SS体系对SDZ的降解率可达95.13%,远高于PI/SS、TiO2/SS和TiO2/PI体系对SDZ的降解效果。
2)协同体系有效作用的溶液pH范围较宽;在目标物的降解过程中,COD不断下降。
3)·O2−是PI/TiO2/SS体系中降解SDZ的最主要的活性物质。
4) PI/TiO2/SS体系对CIP、TMP以及EFA也有较好的降解,说明PI/TiO2/SS协同体系具有高效降解难降解有机物的能力,在有机废水处理中具有一定的应用潜力。
模拟太阳光下高碘酸钠/TiO2协同降解水体抗生素
Synergetic degradation of antibiotics in water by periodate/TiO2 under stimulated solar irradiation
-
摘要: 为了拓宽抗生素废水高效降解的方法,研究采用氙灯作为模拟太阳光源(Simulated sunlight,SS)活化高碘酸盐(Periodate,PI),建立用于抗生素降解的水处理体系,并通过在其中引入TiO2(PI/TiO2/SS),提升作用效果。研究首先以磺胺嘧啶(sulfadiazine,SDZ)为代表,分析了PI/TiO2/SS系统中影响其降解的关键参数,包括TiO2含量、PI含量、SDZ质量浓度和溶液初始pH等。结果表明,在0.3 mmol·L−1 PI和0.05 g·L−1 TiO2的条件下,处理30 min后SDZ的去除率可达95.13%,且PI含量越高、TiO2用量越大以及SDZ初始质量浓度越低时,降解率越高;在较宽的溶液pH范围内,PI/TiO2/SS体系均能实现SDZ的高效降解;反应过程中SDZ溶液的UV-Vis光谱和COD去除分析结果证明了PI/TiO2/SS体系协同作用。通过溶液中H2O2质量浓度的测定以及淬灭剂实验揭示了PI/TiO2/SS体系中·O2−>IO3·和·OH>h+对SDZ的降解作用。此外,对比考察了TiO2添加前后甲氧苄啶(Trimethoprim,TMP)、环丙沙星(Ciprofloxacin,CIP)和恩诺沙星(Enrofloxacin,EFA)溶液降解效果的变化,在一定程度上证明了PI/TiO2/SS体系针对抗生素废水处理的普适性。Abstract: In order to broaden the efficient degradation method of antibiotics in wastewater, the xenon lamp was used as the simulated sunlight (SS) source to activate the periodate (PI), and the water treatment system for antibiotics degradation was set up accordingly. The TiO2 was introduced to the system to improve the degradation effect. At first, sulfadiazine (SDZ) was taken as a representative, the key parameters in PI/TiO2/SS system affecting SDZ degradation were analyzed, which included TiO2 content, PI content, SDZ concentration and initial pH values of the solutions. The results showed that the SDZ degradation rate could reach 95.13% after 30 min treatment at 0.3 mmol·L−1 PI and 0.05 g·L−1 TiO2. The higher PI content, the higher TiO2 dosage, and the lower the SDZ concentration, the higher the degradation rate. The efficient degradation of SDZ in the PI/TiO2/SS system could be achieved in a wide range of solution pH. The UV-Vis spectra and COD removal analysis of SDZ solution during the reaction process proved the synergetic degradation effect of PI/TiO2/SS system. The action rule of ·O2− > IO3· and ·OH > h+ in the PI/TiO2/SS system for the SDZ degradation was revealed through the experiments of the H2O2 concentration detection and quenching experiments. In addition, the study investigated and compared the degradation of trimethoprene (TMP), ciprofloxacin (CIP) and enrofloxacin (EFA) solutions before and after TiO2 addition, which proved the universality of PI/TiO2/SS system for the treatment of antibiotic wastewater to some extent.
-
Key words:
- simulated sunlight /
- TiO2 /
- periodate activation /
- antibiotic degradation /
- mechanism analysis
-
-
表 1 正交实验设计
Table 1. Orthogonal experimental design
因素 水平 (A )TiO2/(g·L−1) (B) PI/(mmol·L−1) (C) pH 1 0.05 0.1 5 2 0.10 0.2 7 3 0.15 0.3 9 表 2 正交实验结果
Table 2. Orthogonal experimental results
序号 影响因子 A B C 降解率/% 1 0.05 0.1 5 57.72 2 0.05 0.2 7 82.71 3 0.05 0.3 9 94.97 4 0.10 0.1 7 60.1 5 0.10 0.2 9 83.4 6 0.10 0.3 5 94.1 7 0.15 0.1 9 61.75 8 0.15 0.2 5 83.88 9 0.15 0.3 7 95.46 K1 235.40 179.57 235.70 K2 237.60 249.99 238.27 K3 241.09 284.53 240.12 k1 78.47 59.86 78.57 k2 79.20 83.33 79.42 k3 80.36 94.84 80.04 R 1.90 34.99 1.47 -
[1] ZHANG Q Q, YING G G, PAN C G, et al. Comprehensive evaluation of antibiotics emission and fate in the river basins of China: source analysis, multimedia modeling, and linkage to bacterial resistance[J]. Environmental Science & Technology, 2015, 49(11): 6772-6782. [2] 俞馨贤. Bi2WO6光催化剂制备改性及光催化降解磺胺嘧啶性能研究[D]. 西藏: 西藏大学, 2023. [3] 杨佳妮. 基于柠檬酸铁的Fe/C催化剂制备及其活化过硫酸盐降解磺胺嘧啶的性能研究[D]. 甘肃: 兰州交通大学, 2023. [4] JIA A, WAN Y, XIAO Y, et al. Occurrence and fate of quinolone and fluoroquinolone antibiotics in a municipal sewage treatment plant[J]. Water Research, 2012, 46(2): 387-394. doi: 10.1016/j.watres.2011.10.055 [5] VALIALO P, KRUGLOVA A, MIKOLA A, et al. Toxicological impacts of antibiotics on aquatic micro-organisms: a mini-review[J]. International Journal of Hygiene and Environmental Health, 2017, 220(3): 558-569. doi: 10.1016/j.ijheh.2017.02.003 [6] Han N, Wang S, Yao Z, et al. Superior three‐dimensional perovskite catalyst for catalytic oxidation[J]. EcoMat, 2020, 2(3): e12044. doi: 10.1002/eom2.12044 [7] ALHARBI S K, KANG J, NGHIEM L D, et al. Photolysis and UV/H2O2 of diclofenac, sulfamethoxazole, carbamazepine, and trimethoprim: Identification of their major degradation products by ESI–LC–MS and assessment of the toxicity of reaction mixtures[J]. Process Safety and Environmental Protection, 2017, 112: 222-234. doi: 10.1016/j.psep.2017.07.015 [8] LIU Z, DEMEESTERE K, VAN H S. Comparison and performance assessment of ozone-based AOPs in view of trace organic contaminants abatement in water and wastewater: a review[J]. Journal of Environmental Chemical Engineering, 2021, 9(4): 105599. doi: 10.1016/j.jece.2021.105599 [9] QIN W, FANG G, WANG Y, et al. Mechanistic understanding of polychlorinated biphenyls degradation by peroxymonosulfate activated with CuFe2O4 nanoparticles: Key role of superoxide radicals[J]. Chemical Engineering Journal, 2018, 348: 526-534. doi: 10.1016/j.cej.2018.04.215 [10] NIU L, ZHANG K, JIANG L, et al. Emerging periodate-based oxidation technologies for water decontamination: A state-of-the-art mechanistic review and future perspectives[J]. Journal of Environmental Management, 2022, 323: 116241. doi: 10.1016/j.jenvman.2022.116241 [11] DAI J, WANG Z, CHEN K, et al. Applying a novel advanced oxidation process of biochar activated periodate for the efficient degradation of bisphenol A: Two nonradical pathways[J]. Chemical Engineering Journal, 2023, 453: 139889. doi: 10.1016/j.cej.2022.139889 [12] ZONG Y, ZHANG H, SHAO Y, et al. Surface-mediated periodate activation by nano zero-valent iron for the enhanced abatement of organic contaminants[J]. Journal of Hazardous Materials, 2022, 423: 126991. doi: 10.1016/j.jhazmat.2021.126991 [13] DU J, TANG S, LING H, et al. Insights into periodate oxidation of bisphenol a mediated by manganese[J]. Chemical Engineering Journal, 2019, 369: 1034-1039. doi: 10.1016/j.cej.2019.03.158 [14] LING C, WU S, HAN J, et al. Sulfide-modified zero-valent iron activated periodate for sulfadiazine removal: Performance and dominant routine of reactive species production[J]. Water Research, 2022, 220: 118676. doi: 10.1016/j.watres.2022.118676 [15] CHOI Y, YOON H I, LEE C, et al. Activation of periodate by freezing for the degradation of aqueous organic pollutants[J]. Environmental Science & Technology, 2018, 52(9): 5378-5385. [16] LEE Y C, CHEN M J, HUANG C P, et al. Efficient sonochemical degradation of perfluorooctanoic acid using periodate[J]. Ultrasonics Sonochemistry, 2016, 31: 499-505. doi: 10.1016/j.ultsonch.2016.01.030 [17] ZHANG X, YU X, YU X, et al. Efficiency and mechanism of 2, 4-dichlorophenol degradation by the UV/IO4− process[J]. Science of the Total Environment, 2021, 782: 146781. doi: 10.1016/j.scitotenv.2021.146781 [18] CHIA L H, TANG X, WEAVERS L K. Kinetics and mechanism of photoactivated periodate reaction with 4-chlorophenol in acidic solution[J]. Environmental Science & Technology, 2004, 38(24): 6875-6880. [19] DJABALLH M L, MEROUANI S, BENDJAMA H, et al. Development of a free radical-based kinetics model for the oxidative degradation of chlorazol black in aqueous solution using periodate photoactivated process[J]. Journal of Photochemistry and Photobiology A: Chemistry, 2021, 408: 113102. doi: 10.1016/j.jphotochem.2020.113102 [20] SUN H, HE F, CHOI W. Production of reactive oxygen species by the reaction of periodate and hydroxylamine for rapid removal of organic pollutants and waterborne bacteria[J]. Environmental Science & Technology, 2020, 54(10): 6427-6437. [21] BOKARE A D, CHOI W. Singlet-oxygen generation in alkaline periodate solution[J]. Environmental Science & Technology, 2015, 49(24): 14392-14400. [22] ZONG Y, SHAO Y, ZENG Y, et al. Enhanced oxidation of organic contaminants by iron (II)-activated periodate: the significance of high-valent iron–oxo species[J]. Environmental Science & Technology, 2021, 55(11): 7634-7642. [23] LONG Y, DAI J, ZHAO S, et al. Atomically dispersed cobalt sites on graphene as efficient periodate activators for selective organic pollutant degradation[J]. Environmental Science & Technology, 2021, 55(8): 5357-5370. [24] DU J, XIAO G, XI Y, et al. Periodate activation with manganese oxides for sulfanilamide degradation[J]. Water Research, 2020, 169: 115278. doi: 10.1016/j.watres.2019.115278 [25] LONG Y, DAI J, ZHAO S, et al. Metal–organic framework-derived magnetic carbon for efficient decontamination of organic pollutants via periodate activation: surface atomic structure and mechanistic considerations[J]. Journal of Hazardous Materials, 2022, 424: 126786. doi: 10.1016/j.jhazmat.2021.126786 [26] WANG Q, ZENG H, LIANG Y, et al. Degradation of bisphenol AF in water by periodate activation with FeS (mackinawite) and the role of sulfur species in the generation of sulfate radicals[J]. Chemical Engineering Journal, 2021, 407: 126738. doi: 10.1016/j.cej.2020.126738 [27] ZHANG X, KAMALI M, ULENERS T, et al. UV/TiO2/periodate system for the degradation of organic pollutants–kinetics, mechanisms and toxicity study[J]. Chemical Engineering Journal, 2022, 449: 137680. doi: 10.1016/j.cej.2022.137680 [28] LIU F, LI Z, DONG Q, et al. Catalyst-free periodate activation by solar irradiation for bacterial disinfection: performance and mechanisms[J]. Environmental Science & Technology, 2022, 56(7): 4413-4424. [29] GUO R, QI Y, LI B, et al. Efficient degradation of alkyl imidazole ionic liquids in simulated sunlight irradiated periodate system: Kinetics, reaction mechanisms, and toxicity evolution[J]. Water Research, 2022, 226: 119316. doi: 10.1016/j.watres.2022.119316 [30] HUANG Y, XIE Q, WANG H, et al. Degradation of trimethoprim in the simulated solar light/periodate system: Process and mechanism analysis[J]. Journal of Water Process Engineering, 2024, 57: 104726. doi: 10.1016/j.jwpe.2023.104726 [31] MAO D, YAN X, WANG H, et al. Catalysis of rGO-WO3 nanocomposite for aqueous bisphenol a degradation in dielectric barrier discharge plasma oxidation process[J]. Chemosphere, 2021, 262: 128073. doi: 10.1016/j.chemosphere.2020.128073 [32] CHEN M, CHU W. Photo-oxidation of an endocrine disrupting chemical o-chloroaniline with the assistance of TiO2 and iodate: Reaction parameters and kinetic models[J]. Chemical Engineering Journal, 2014, 248: 273-279. doi: 10.1016/j.cej.2014.03.038 [33] LI R, WANG J, WU H, et al. Periodate activation for degradation of organic contaminants: Processes, performance and mechanism[J]. Separation and Purification Technology, 2022, 292: 120928. doi: 10.1016/j.seppur.2022.120928 [34] LI X, GAN X. Photo-Fenton degradation of multiple pharmaceuticals at low concentrations via Cu-doped-graphitic carbon nitride (g-C3N4) under simulated solar irradiation at a wide pH range[J]. Journal of Environmental Chemical Engineering, 2022, 10(5): 108290. doi: 10.1016/j.jece.2022.108290 [35] WEAVERS L K, HUA I, HOFFMANN M R. Degradation of triethanolamine and chemical oxygen demand reduction in wastewater by photoactivated periodate[J]. Water Environment Research, 1997, 69(6): 1112-1119. doi: 10.2175/106143097X125849 [36] ZHU Y, WANG F, ZHOU B, et al. Photo-assisted Fe2+ modified molybdenum disulfide activated potassium persulfate to degrade sulfadiazine: Insights into the degradation pathway and mechanism from density functional theory[J]. Chemical Engineering Journal, 2022, 435: 134904. doi: 10.1016/j.cej.2022.134904 [37] YAO J, DONG Z, YE X, et al. Electrochemically activated peroxymonosulfate with mixed metal oxide electrodes for sulfadiazine degradation: Mechanism, DFT study and toxicity evaluation[J]. Chemosphere, 2022, 309: 136695. doi: 10.1016/j.chemosphere.2022.136695 [38] KINNEY C A, FURLONG E T, ZAUGG D, et al. Survey of organic wastewater contaminants in biosolids destined for land application[J]. Environmental Science & Technology, 2006, 40(23): 7207-7215. [39] FENG Y, SHIH K. Sulfate radical-mediated degradation of sulfadiazine by CuFeO2 rhombohedral crystal-catalyzed peroxymonosulfate: Synergistic effects and mechanisms[J]. Environmental Science & Technology, 2016, 50: 3119-3127. -


 下载:
下载: